Keywords
rivaroxaban, bullous pemphigoid-like, cutaneous drug eruptions
Abstract
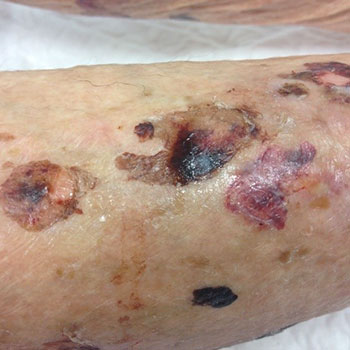
Little has been documented about hypersensitivity reactions caused by treatment with rivaroxaban. This paper reports a bullous pemphigoid-like skin eruption that occurred in a 76-year-old female patient during rivaroxaban treatment. This case highlights the vigilance required by healthcare workers in recognising potential adverse effects of newly marketed drugs and in making medication changes when necessary. A bullous pemphigoid-like eruption due to treatment with rivaroxaban has not, to the best of the Authors’ knowledge, been reported previously in the literature.
References
Views: 1647
HTML downloads: 2432
PDF downloads: 803
Published:
2018-01-15
Issue:
Vol. 5 No. 3
(view)










