Keywords
Pulmonary fibrosis, COVID-19, anakinra
Abstract
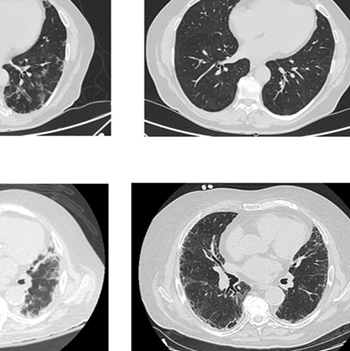
Coronavirus disease 2019 (COVID-19) is currently a major public health problem. The development of pulmonary fibrosis secondary to acute respiratory distress syndrome (ARDS) is one of the expected sequelae. In this case series, we describe five instances of the use of anakinra in late-phase COVID-19 pneumonia in hospitalized patients with pulmonary fibrosis and refractory respiratory failure fulfilling ARDS criteria. The study demonstrates that anakinra has promising efficacy and safety in late-phase COVID-19 infection in patients with ARDS and refractory hypoxaemia, and suggests its potential application as antifibrotic therapy in these patients.
References
Views: 931
HTML downloads: 74
PDF downloads: 298
Published:
2021-10-08
Issue:
2021: Vol 8 No 10
(view)










