ABSTRACT
May–Thurner syndrome (MTS) usually presents as acute or chronic deep vein thrombosis (DVT) in patients. A 49-year-old woman presented with left lower limb DVT, which was followed by a diagnosis of MTS on a background of polycystic ovary syndrome (PCOS) and hypothyroidism. MTS is more common among women in the second to fourth decades of life. An endovascular approach is the preferred first-line treatment for MTS.
LEARNING POINTS
- This case highlights the importance of serial ultrasound Doppler scans in cases where the probability of deep vein thrombosis (DVT) is high as a negative initial Doppler scan does not exclude DVT.
- May–Thurner syndrome (MTS) should be suspected especially in younger patients in whom no cause for DVT has been found.
- Missing a diagnosis of MTS could result in life-threatening conditions such as pulmonary embolism and post-thrombotic syndrome.
KEYWORDS
Deep vein thrombosis, May–Thurner syndrome, endovascular treatment
INTRODUCTION
In 1851, Virchow was the first to observe that iliofemoral deep vein thromboses (DVTs) are five times more common in the left limb compared with the right. In 1957, May and Thurner explained this finding as due to an anatomical variation of the left common iliac vein. They found that a ‘venous spur’ (an intraluminal thickening), caused by the chronic pulsation of the right common iliac artery, caused external compression of the left common iliac vein which eventually resulted in venous outflow obstruction[1].
Although the true prevalence of May–Thurner syndrome (MTS) is unknown, it is estimated to be present in over 20% of the population. However, only 2–3% of all lower limb DVTs are related to MTS[1].
CASE DESCRIPTION
A 49-year-old woman presented with a 6-hour history of swelling of the left lower limb (which was 5 cm greater in diameter than the right), calf tenderness, and pitting oedema associated with pain radiating from the groin down to the foot which worsened on weight bearing. The patient had a Wells DVT probability score of 4. Warm and palpable pulses were noted in both lower limbs. The rest of the clinical examination was unremarkable and vital signs were stable. The patient’s past medical history included polycystic ovary syndrome (PCOS) and hypothyroidism. She was a non-smoker with occasional alcohol intake. Her father had died of a myocardial infarction, but there was no family history of clotting disorders. Her medications on admission were Eltroxin 50 μg and an oral contraceptive pill. Prior to admission she was mobilising actively and independently.
Her WBC count was 14.5, neutrophils 11.62, D-dimer 7.56 mg/l and CRP 56.7 mg/l.
Coagulation, renal profile and a repeat full blood count were normal. A repeat CRP was 132 mg/l, LDH 403 U/l, total protein 59 g/l and albumin 30 g/l. A thrombophilia screen was negative.
The patient was treated with a therapeutic dose of Innohep for a probable left lower limb DVT, and benzylpenicillin 2.4 g four times a day iv and flucoxacillin 2 g four times a day iv for thrombophlebitis.
A US Doppler scan of the left lower limb was negative for DVT so the therapeutic dose of Innohep was changed to a prophylactic dose. A repeat US Doppler scan of the left lower limb after 48 hours (Figs. 1 and 2a) revealed an extensive left leg DVT extending up into the left iliac vein.
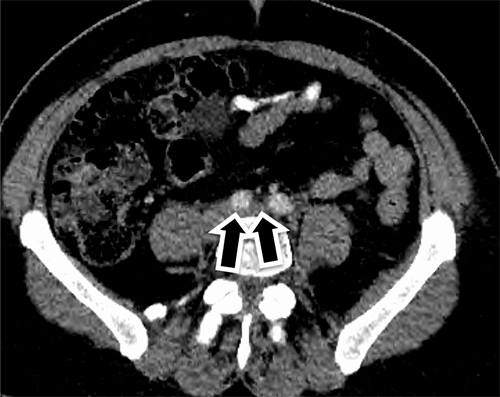
Figure 1. Axial CT scan of the pelvis demonstrates a compression of the left common iliac vein (long arrow) as it passes under the right common iliac artery (arrow head)
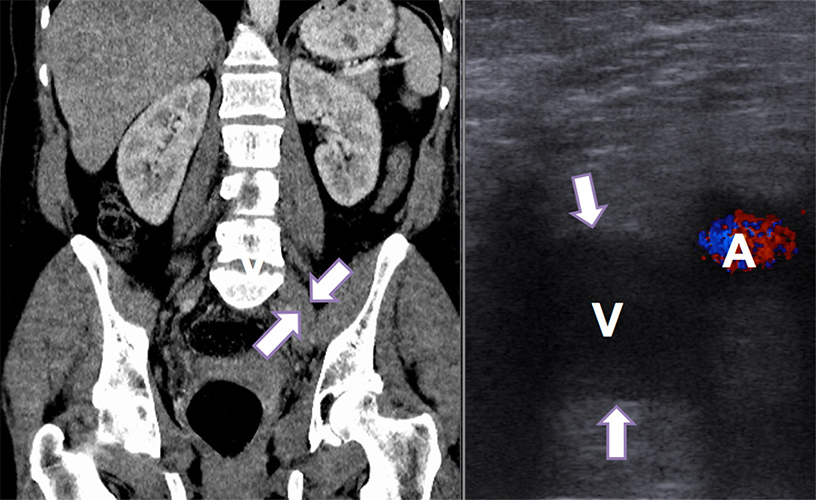
Figure 2 a, b. Coronal CT scan and US images demonstrate a DVT in the left external iliac vein (long arrow)
A CT scan of the thorax, abdomen and pelvis (Figs. 2b and 3) showed that the left common iliac vein was compressed against the lumbar vertebral body and the overlying right common artery with evidence of a DVT in the left lower limb, indicating May–Thurner syndrome.
The prophylactic dose of Innohep was changed back to a therapeutic dose. However, Innohep was stopped after 2 days. The patient was commenced on apixaban 10 mg twice a day for a week and then 5 mg twice a day for 6 months and wore grade 2 compression stockings. She was discharge after 8 days with vascular team follow-up scheduled for 1 week later and outpatient follow-up for 3 weeks later.
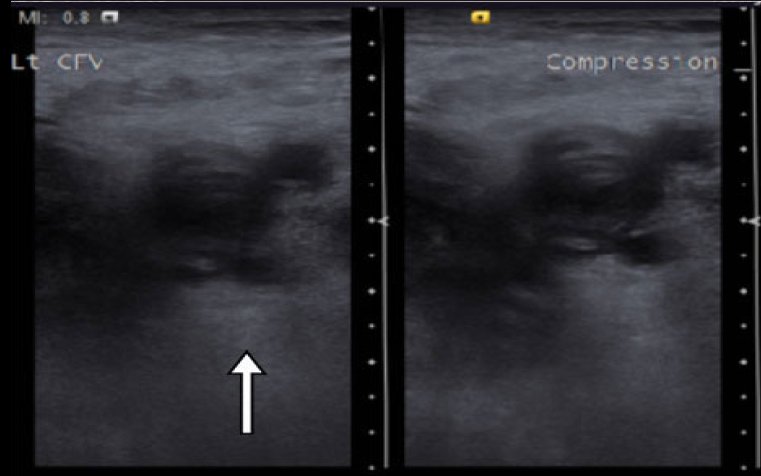
Figure 3. US images with and without compression demonstrate a DVT in the left femoral vein (long arrow)
The patient was reviewed by the vascular team 1 week after discharge. Anticoagulation was continued and therapeutic Innohep commenced. A CT venogram showed a left ileofemoral DVT. The patient was readmitted to hospital. Access was gained via the internal jugular vein and an infrarenal inferior vena cava (IVC) filter was placed.
Mechanical thrombolectomy was performed and the patient was started on thrombolysis with alteplase 20 mg diluted in normal saline. However, the patient became hypotensive after approximately 15 min of infusion. The procedure was suspended and the patient was started on iv hydrocortisone and fluid resuscitation. A CT of the abdomen and pelvis including the lower limbs ruled out haematoma and haemorrhage. The patient became clinically stable.
During her admission the patient was also diagnosed with delayed cell-mediated-type cutaneous hypersensitivity reaction caused by the iohexol (Omnipaque) used during radiological imaging. She was treated with betnovate cream, Piriton and steroids. No anaphylactic-type reaction was noted and the patient was clinically stable.
After nearly 2 weeks the patient was discharged from hospital on therapeutic Innohep. A skin patch test was carried out and the infrarenal IVC filter was removed a few weeks later.
DISCUSSION
When the pre-test probability suggests DVT, D-dimer is positive and initial simplified compression ultrasonography is negative, serial ultrasonography tests should be carried out after 48 hours and/or after 7 days[2].
MTS usually presents as acute or chronic DVT, which may result in pulmonary embolism and symptoms of chronic venous outflow obstruction, including unilateral left lower limb swelling, pain, claudication, venous stasis ulceration and varicosities[1].
The gold standard method for diagnosis of MTS is digital subtraction venography obtained via femoral access as both compression of the vein and the presence of venous collaterals joining contralateral veins can be detected. Other methods used include colour Doppler ultrasonography (US), magnetic resonance angiography (MRV) and computerized tomography venography (CRV). However, colour Doppler US cannot evaluate vein compression accurately, and although MRV and CTV are both non-invasive imaging tools that can detect anatomical structures, intraluminal septation resulting in venous flow obstruction may not be clearly visible[3].
DVT is usually only treated if it is symptomatic[1]. Although anticoagulation is the mainstay of treatment to prevent thrombus propagation, it is inadequate for an existing chronic thrombus, as it does not dissolve it, which may lead to complications such as recurrence of thrombosis and post-thrombotic syndrome[4].
Revascularization is the definitive treatment in symptomatic MTS. Endovascular therapy includes catheter-directed thrombolysis, percutaneous mechanical thrombectomy, percutaneous balloon venoplasty and stenting[4]. Following stent insertion, the patient is treated with anticoagulants for at least 6 months to prevent in-stent stenosis[1]. Insertion of an IVC filter before any lower limb interventions is recommended to prevent pulmonary events.
In catheter-directed thrombolysis (CDT), the catheter is often left in place for 48 hours or longer so that the risk of bleeding can be lowered by administering a lower dose for a shorter duration of time. CDT together with percutaneous mechanical thrombectomy has proven to be much more effective than CDT alone in recent treatment regimes, mostly in acute rather than chronic thrombosis[4].
Percutaneous balloon venoplasty followed by stenting has advantages such as the prevention of early venous re-occlusion and increased primary patency rates. The added benefit of using a larger stent is that it reduces the risk of migration[4].
In chronic iliofemoral occlusion, where CDT cannot be considered, surgical intervention is advised. The procedure includes vein patch angioplasty, intraluminal band excision, division and relocation of the right common iliac artery behind the left common femoral vein, and creation of a vein graft bypass between the contralateral saphenous and ipsilateral common femoral veins where a temporary arteriovenous fistula is created[5].