ABSTRACT
Persistent left superior vena cava (PLSVC) is a common congenital anomaly of the thoracic venous system. It is usually asymptomatic and is detected when cardiovascular imaging is performed for unrelated reasons. Its presence can complicate catheter placement within the right side of the heart, as well as cardiac procedures. In most cases PLSVC can go unnoticed and only becomes pertinent when patients require cardiac imaging or surgery. Once found, it puts patients at risk for cardiac interventions. Here we present a case that highlights the practical implications of PLSVC where a patient developed flash pulmonary oedema after gastrointestinal procedures – in this case, laparoscopic cholecystectomy – as well as endoscopic retrograde cholangiopancreatography (ERCP). Management is also discussed, leading to the resolution of symptoms. The report will also propose a pathophysiologic mechanism leading to this complication that is not well understood in the medical literature.
LEARNING POINTS
- Complications of gastrointestinal procedures are not studied in patients with persistent left superior vena cava syndrome.
- Inform the medical community of the possible pathophysiology of flash pulmonary oedema after gastrointestinal manipulation in patients with persistent left superior vena cava syndrome.
KEYWORDS
Internal medicine, gastroenterology, cardiology, critical care, Persistent left superior vena cava, PLSVC
INTRODUCTION
Persistent left superior vena cava (PLSVC) is a common venous anomaly that is found in 0.5% of the normal population[1]; it develops in early foetal life and is characterised by the persistence of the left anterior cardinal vein leading to PLSVC. In 80–90% of cases, the PLSVC drains into the right atrium via the coronary sinus[2]. It begins at the junction of the left subclavian and internal jugular veins and passes through the left side of the mediastinum adjacent to the arcus aorta[4]. In the remainder of the cases, it enters directly or through the pulmonary veins into the left atrium[3].
CASE DESCRIPTION
The patient is an 86-year-old female with a past medical history of paroxysmal atrial fibrillation, hypertension, cataracts, osteoporosis and hypothyroidism. She presented to a community hospital with stomach and back pain, which began the day before. The pain was localised to the epigastric region, radiating to the back; it was rated 10/10 in intensity, and not relieved with acetaminophen. She also complained of nonbilious, non-bloody vomiting. The patient had nausea and loss of appetite throughout the day and has never had similar symptoms in the past.
On admission, the patient had a CT scan of the abdomen and pelvis with contrast, which revealed multiple large gallstones in the gallbladder and fluid surrounding the pancreas with associated inflammation. Blood lipase was elevated at >3500 U/l (reference 12–53), lactic acid levels were also elevated at 1.93 mmol/l (reference 0.4–1.9), elevated total bilirubin was 1.7 mg/dl (reference 0–1) and elevated alkaline phosphatase was 162 U/l (reference 50–138). The patient was diagnosed with pancreatitis and was started on IV fluids and pain control. Lipase levels were monitored and eventually normalised, leading to resolution of the pancreatitis. The patient continued to complain of abdominal pain now located in the right upper quadrant; sharp, with worsening liver function tests and alkaline phosphatase. General surgery was consulted, and the patient was recommended a laparoscopic cholecystectomy. She received an echocardiogram as part of the cardiac clearance, which revealed a normal systolic function with an ejection fraction of 50–55%. It also showed a dilated coronary sinus with PLSVC terminating in the left atrium, with atrial septal bowing from left to right consistent with increased left atrial pressure. The findings were communicated to the patient; she was informed of the cholecystectomy surgical risks which included bile leak, incisional hernia and intra-abdominal abscess[9] as well as risks seen in cardiac dysfunction, which included hypertension, tachyarrhythmia and bradycardia[10]. The patient agreed to proceed with the surgery.
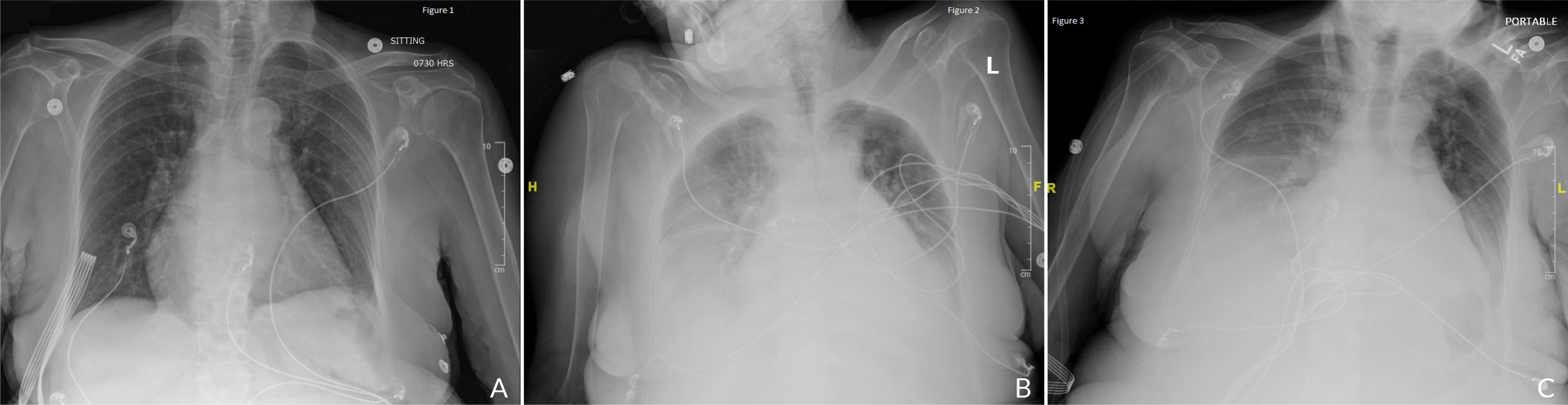
The patient underwent a laparoscopic cholecystectomy the following day and was transferred to the recovery unit after extubation in a haemodynamically stable condition. At night the patient started developing hypoxia and tachycardia, and her oxygen started desaturating to the 70s, so she was put on a high-flow nasal cannula. A chest X-ray revealed diffuse bilateral interstitial and airspace opacities with bilateral pleural effusions (Fig. 1). The patient was started on furosemide, which resolved her pulmonary oedema.
Overnight, the abdominal pain continued to persist. At that point, the patient received a magnetic resonance cholangiopancreatography which revealed choledocholithiasis with two stones; one in the mid-common bile duct and the second more distally in the common bile duct. At that point, Gastroenterology was consulted and the patient was recommended an endoscopic retrograde cholangiopancreatography (ERCP), which she agreed to. Soon after the ERCP, the patient experienced hypoxia and was admitted to the ICU for observation, and was put on bilevel-positive airway pressure. A chest X-ray revealed bilateral pleural effusions (Fig. 1C); the patient was started on furosemide and her condition quickly improved. She was downgraded the following day in a stable condition.
The patient was observed for a few days after the procedure; she was started on a clear liquid diet and was advanced to a regular diet. She worked with the physical therapist to get her strength back to baseline, and was discharged home in a clinically and vitally stable condition. She was advised to follow up with her primary care physician and gastroenterologist 1 week after discharge.
Figure 1. (A): Chest X-ray of the patient on arrival. (B): Chest X-ray post cholecystectomy depicting diffuse bilateral interstitial with bilateral pleural effusions. (C): Chest X-ray post ERCP with bilateral pleural effusions
DISCUSSION
This case highlights a novel complication, the development of flash pulmonary oedema after biliary manipulation in patients with PLSVC.
Flash pulmonary oedema is divided into three stages; the first stage is the recruitment and distention of small pulmonary vessels, the second is the interstitial oedema development stage and the third is the alveolar fluid flooding decreasing gas exchange leading to hypoxia and dyspnoea[6].
The patient developed flash pulmonary oedema twice; once after a laparoscopic cholecystectomy, and another after ERCP. This mechanism can be explained by a twofold cause; first, the post-surgical atelectatic changes in the lung and second, increased blood flow to the dilated coronary sinus after biliary manipulation. This exacerbates the demand on the left atrium leading to a backup of blood in the left atrium, causing increased pulmonary vascular congestion.
The increased vascular congestion leads to cardiogenic pulmonary oedema which is characterised by transudation of protein-poor fluid into the pulmonary interstitium as a result of increased left ventricular filling pressure. Fluid transudation is mediated by a rise in pulmonary capillary pressure that results from an increase in pulmonary venous and left atrial pressure without a change in permeability of the endothelial layers of the pulmonary capillaries. This results in filtration of protein-poor fluid across the pulmonary endothelium into the alveolar spaces, causing decreased diffusing capacity, hypoxia and shortness of breath[5].
Bilevel-positive airway pressure is the recommended first-line method of ventilator support in selected patients with acute respiratory failure secondary to cardiogenic pulmonary oedema[6]. The pathophysiology of respiratory failure is due to an increase in extravascular water, a reduction in lung volume and respiratory system compliance, and an increase in airway resistance[7].
The solution in this case was quick escalation of oxygenation with a high-flow nasal cannula after cholecystectomy, and bilevel-positive airway pressure after ERCP, as well as the administration of IV furosemide, which rapidly resolved the issue.
CONCLUSION
Although PLSVC is considered a benign condition, it can lead to life-threatening complications if it is not appropriately managed. More research needs to be conducted to study the mechanism of pulmonary pathology leading to flash pulmonary oedema in the setting of biliary manipulation procedures. Once more research has been conducted, then the underlying mechanism can be understood.