ABSTRACT
Lutembacher’s syndrome is a rare disease defined as the combination of atrial septal defect and mitral stenosis, which relieves the progression of symptoms resulting from mitral stenosis. We present a case of a previously asymptomatic 70-year-old man having Lutembacher’s syndrome with sinus venosus type of interatrial defect associated with partial anomalous pulmonary venous drainage. Our patient’s only complaint was of mild exertional dyspnea, which started six months prior to referral. Clinical suspicion of the combination of defects arose during transthoracic echocardiography. The patient underwent transesophageal echocardiography and multi-slice computed tomography for defect confirmation. The combination of Lutembacher’s syndrome with this complex deformity of pulmonary venous drainage has rarely been described. Such complex congenital defects with multifaceted physiological repercussions can go largely undiagnosed for the patients affected for most of their lifetimes.
LEARNING POINTS
- Despite the complexity of Lutembacher’s syndrome, including the involvement of several deformities and pathological mechanisms of mitral stenosis, sinus venosus type of interatrial defects with partial anomalous return in the pulmonary veins, the multiplicity of mechanisms involved contribute to altered hemodynamics.
- Multiple intersecting mechanisms can further contribute to delaying congenital heart disease presentation. Congenital heart disease remains a plausible differential in the evaluation of elderly patients presenting with exertional dyspnea.
- Adult congenital heart defects can have very late onset for presentation, especially with physiological processes that paradoxically reduce the severity and progression of symptoms.
KEYWORDS
Congenital heart defect, Elderly congenital heart disease, Adult congenital defect, Atrial septal defect, Mitral stenosis, Anomalous pulmonary venous return
INTRODUCTION
Lutembacher’s syndrome (LS) is a rare disease defined as the combination of atrial septal defect (ASD) with mitral stenosis (MS), which relieves the progression of symptoms resulting from mitral stenosis[1]. LS with sinus venosus type of interatrial defects (SVASD) associated with anomalous pulmonary venous drainage has rarely been described in the literature[2-4]. In this paper, we report a case of a previously asymptomatic 70-year-old man who was diagnosed with mitral stenosis with a sinus venosus ASD combined with partial pulmonary vein anomalies.
CASE PRESENTATION
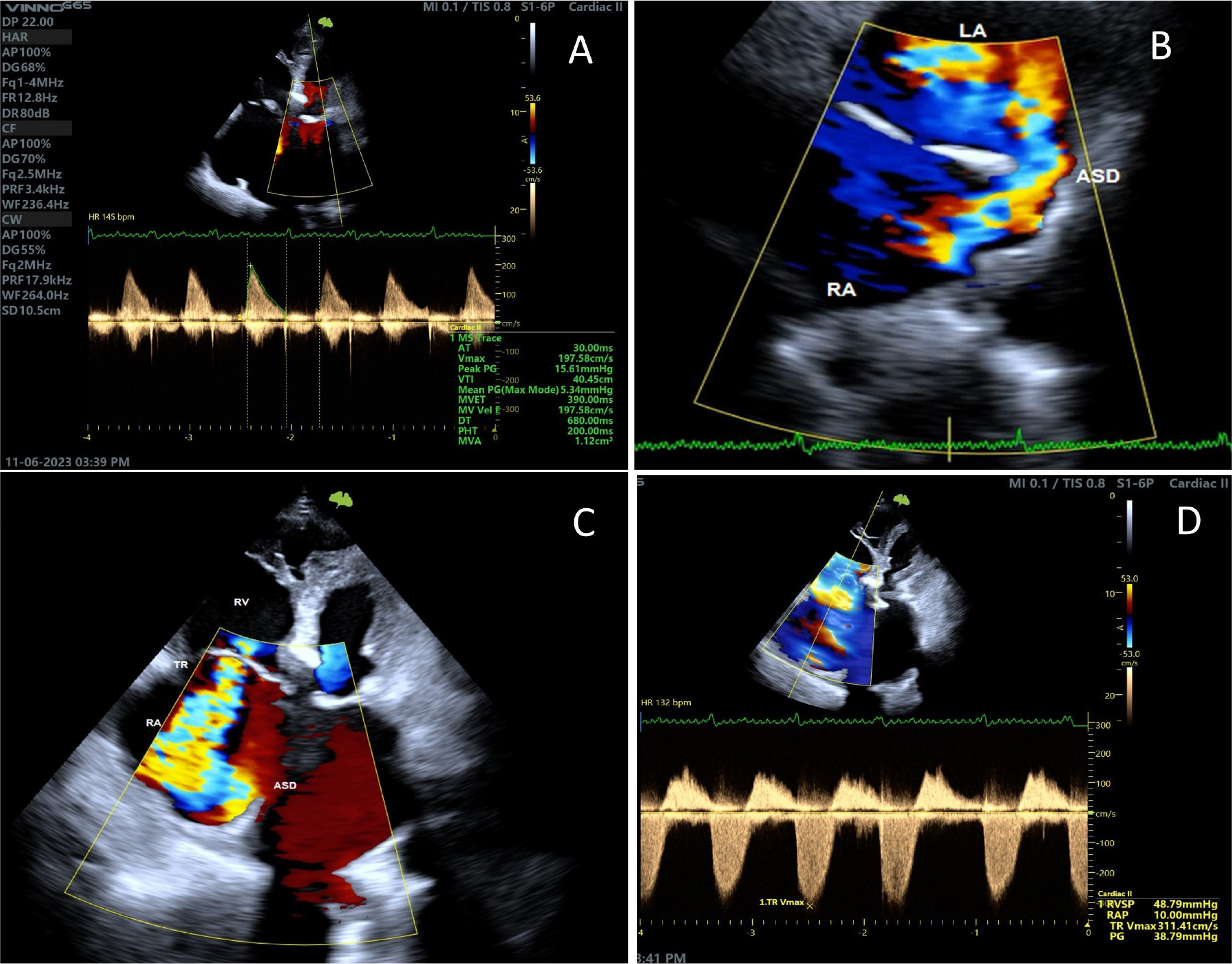
A 70-year-old male was referred to a cardiology outpatient clinic with a complaint of new onset exertional dyspnea (New York Heart Association Class II) for 6 months. The patient denied any chest discomfort or orthopnea, cough, palpitations, or any other cardiac symptoms. On further examination, he reported dyspnea provoked by prolonged or severe exertion for several years, which he attributed to advancing age. He was a 30 pack-year smoker and quit 4 years ago. His past medical history was notable for hypertension, diabetes mellitus, and benign prostatic hyperplasia. His physical examination showed an irregular heart rhythm, grade III/VI holosystolic murmur, and fixed split S2. The rest of the examination was unremarkable. His electrocardiogram showed atrial fibrillation, right bundle branch block, and non-specific ST/T changes. His transthoracic echocardiography showed concentric left ventricle (LV) hypertrophy, moderate LV systolic dysfunction (global ejection fraction = 49%), septal dyskinesis, moderate mitral stenosis (mean pressure gradient = 5.34 mmHg), severe mitral annular calcification with leaflet thickening and diastolic movement restriction accompanied by an SVASD measuring 1.8 cm, and a left to right shunt. The ASD was hemodynamically significant (pulmonary vascular resistance = 2.19 Wood unit and systemic flow ratio (Qp/Qs) = 4.5). An abnormality was found in the drainage of the superior vena cava into the right atrium as the superior vena cava appeared to be strongly dilated. There was also severe tricuspid regurgitation, pulmonary systolic pressure of 49 mmHg, severe bi-atrial enlargement and the right ventricle (RV) was dilated (RV basal diameter = 6.7 cm, RV mid diameter = 6.5 cm Fractional area change (FAC) = 61%, Tricuspid annular plane systolic excursion (TAPSE) = 2.8 cm) (Fig. 1 and 2).
Figure 1. Transthoracic echocardiogram. A) Mitral stenosis gradient; B) ASD color doppler flow; C) Severe tricuspid regurgitation color doppler; D) Tricuspid valve continuous wave doppler signal.
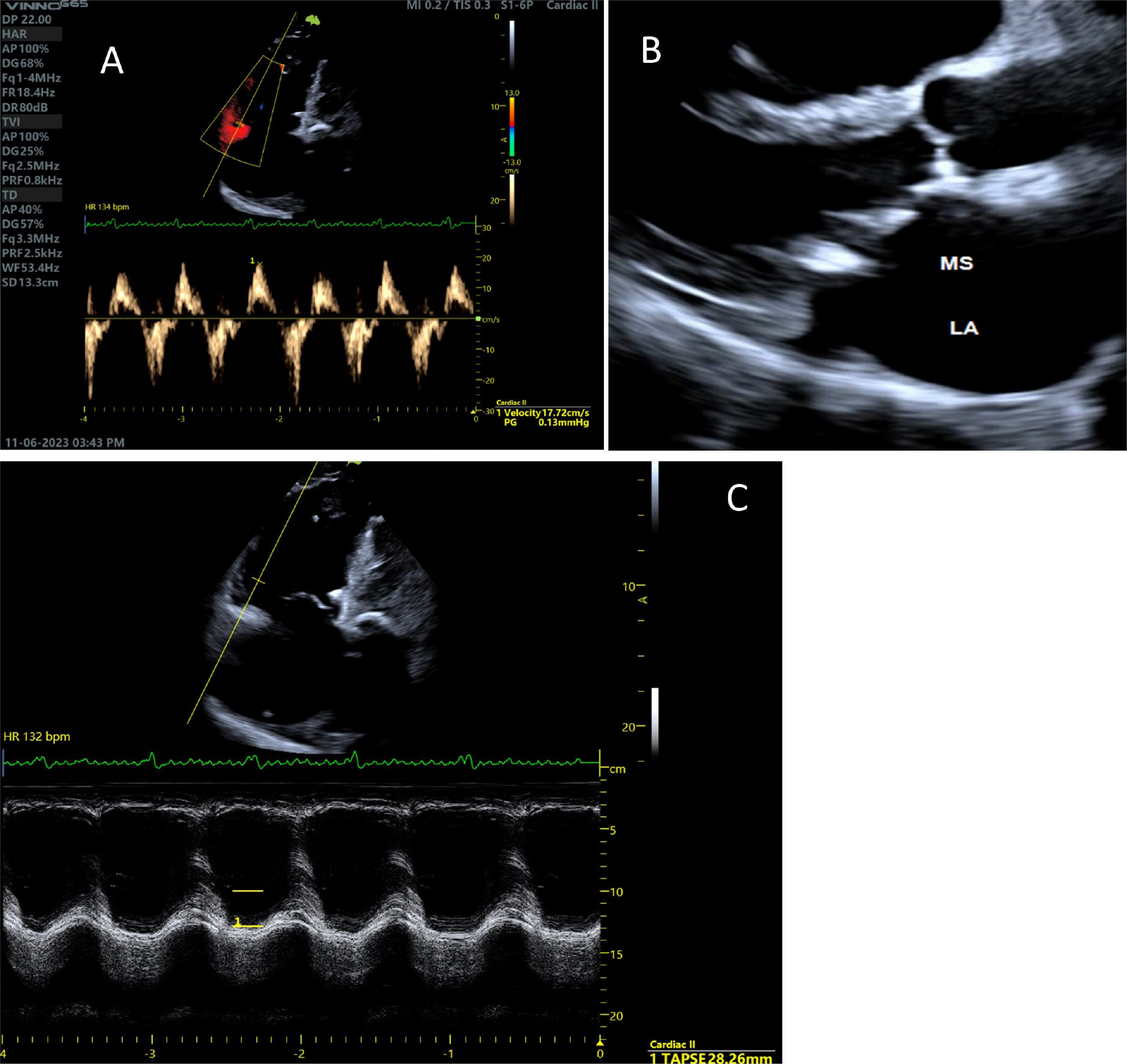
Figure 2. Transthoracic echocardiogram. A) RV free-wall tissue doppler signal; B) Mitral annular calcification in the PLAX view; C) RV TAPSE and apparent right chambers dilatation.
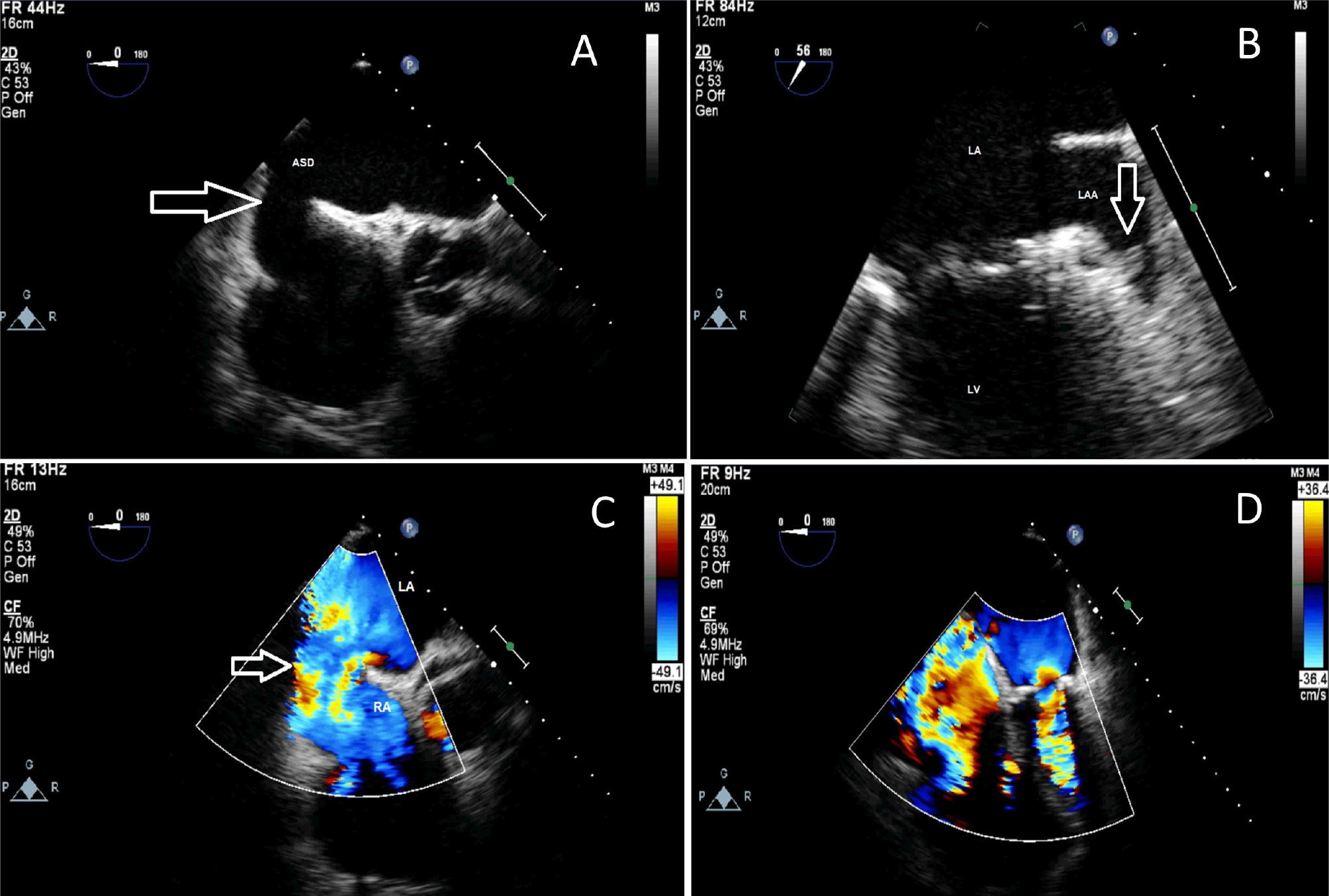
Transesophageal echocardiography showed severe right chamber enlargement with SVASD measuring 1.8 cm. A left to right shunt was noted, in addition to the severe dilatation of the superior vena cava draining into the ASD area, and the innominate vein was readily identifiable. A connecting vein draining into the ASD area was found. There also thinning in the area of the foramen ovale without a visible shunt. The left atrial appendage had irregular echogenic shadows, indicating the presence of thrombi. Moderate mitral stenosis with severe mitral annular calcification, leaflet thickening, and diastolic movement restriction were confirmed (Fig. 3).
Figure 3. Transesophageal echocardiogram. A) ASD location ME AV Q 4-chamber view; B) Left atrial appendage ME left atrial appendage view; C) ASD color doppler signal D. Mitral valve color doppler signal ME 4-chamber view.
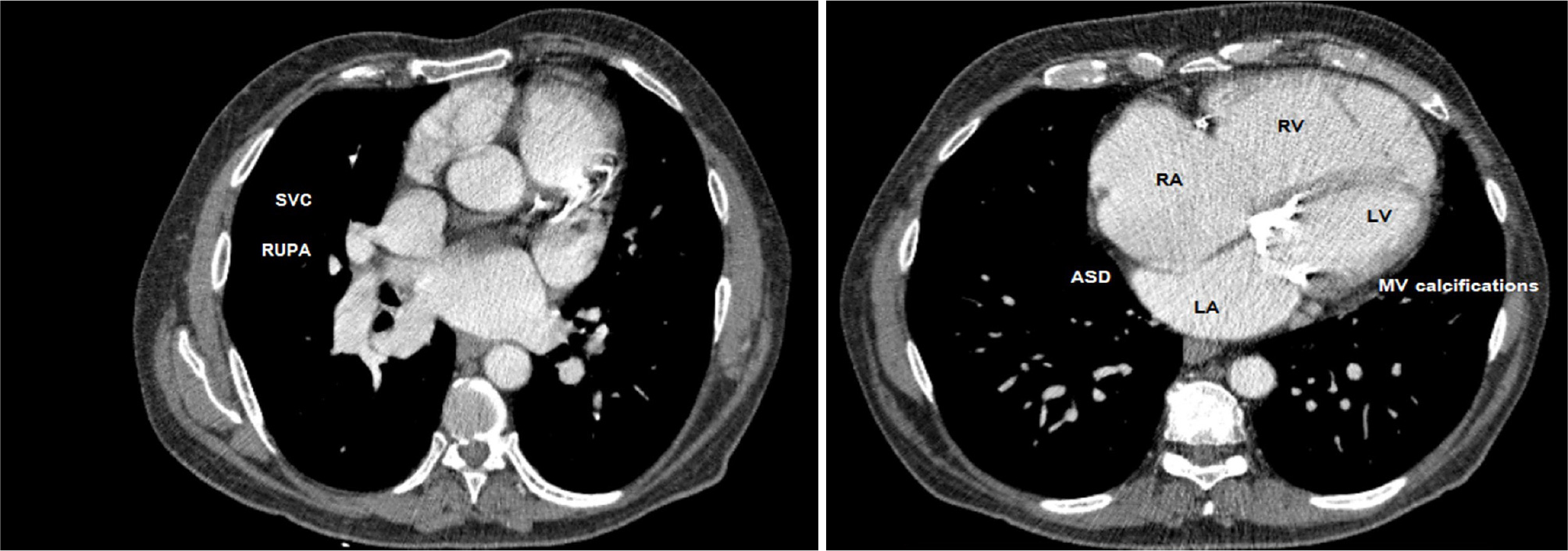
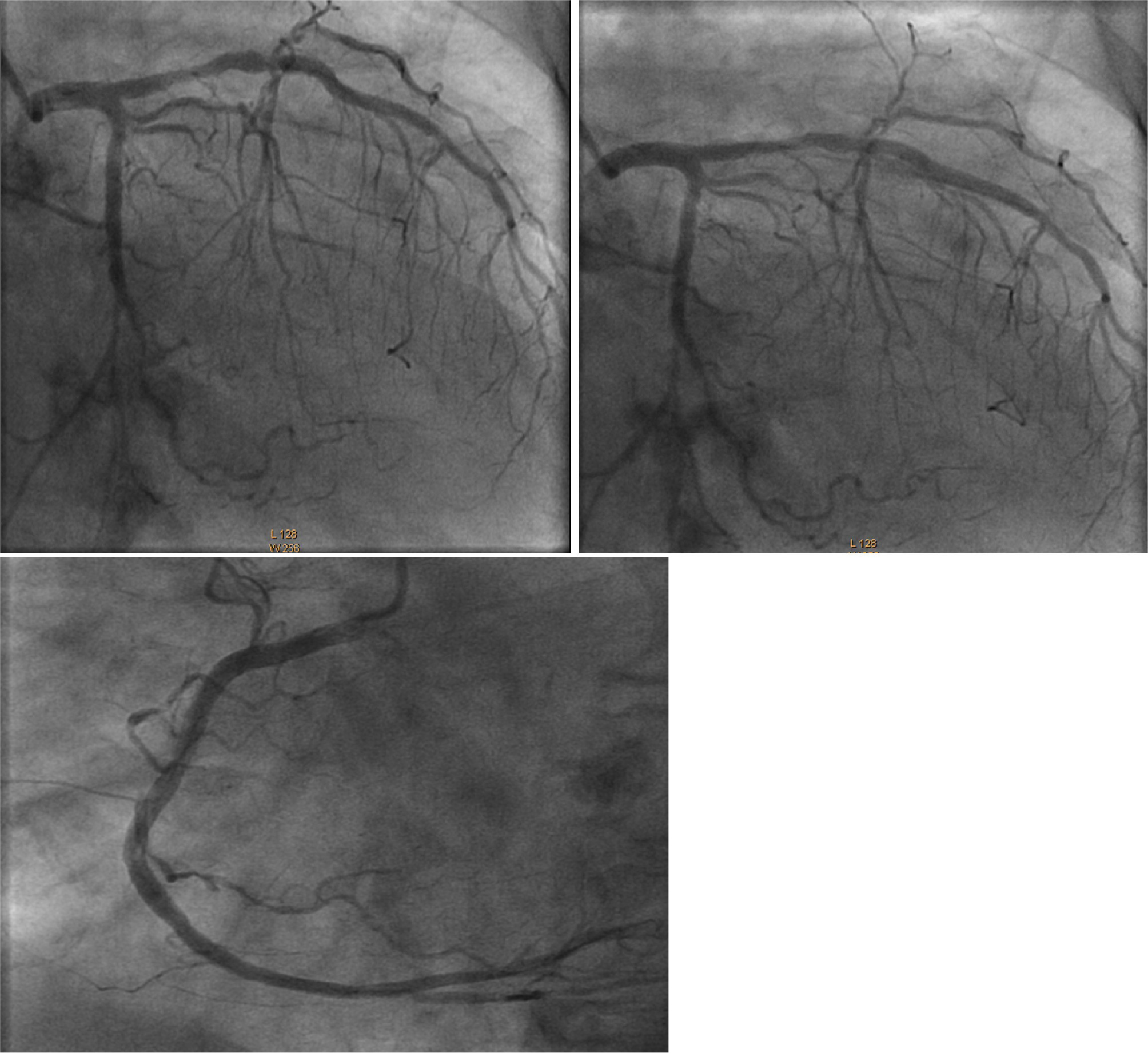
A multi-slice computed tomography scan with contrast was conducted. It revealed a partial anomalous venous return of the right pulmonary vein, where the right upper pulmonary vein was identified as a connecting vein draining directly into the superior vena cava. There was also a small additional inferior pulmonary vein originating from the left lower pulmonary vein also draining into the superior vena cava at the level of the ASD. The superior vena cava and inferior vena cava drained into the right atrium and the innominate vein was also readily distinguishable (Fig. 4). A coronary angiography was performed and showed mild coronary lesions (Fig. 5). Surgical intervention was scheduled.
Figure 4. Multi-slice computed tomography views showing SVC and connecting pulmonary vein.
DISCUSSION
We present a case of a combination of SVASD, mitral stenosis, and partial anomalous pulmonary venous return in an elderly patient. LS is a rare condition referring to the presence of ASD and MS at the same time. Both may be congenital or acquired. Of the ASD types, an ostium secundum is the most common type of ASD in LS, whereas the SVASD type is rare, especially when associated with abnormalities in the drainage of the pulmonary veins[4]. This is the case in our patient. Congenital mitral stenosis is very rare[5]. Our patient’s late presentation could be explained by the acquired etiology of the diagnosed mitral stenosis.
The hemodynamic effects of this abnormality are a result of the interplay between the relative hemodynamic effects of the ASD and mitral stenosis. There are two exits for the flow from the left atrium, the ASD and the stenotic mitral valve. As the blood moves from the left atrium to the right atrium through the ASD, it relieves the pressure within the left atrium, and thus the pulmonary veins. This leads to diminishing pulmonary congestion and symptoms experienced by the patient[6]. This could explain the only moderate limitation of activity that our patient presented with.
The silent presentation for mitral stenosis in the context of LS was also reported in the literature[7]. Conversely, the auscultatory findings of our case were of an isolated holosystolic murmur likely due to tricuspid regurgitation. The large ASD and high left atrial pressure caused by mitral stenosis make the development of Eisenmenger syndrome very rare[8]. Because of this mechanism, the onset of symptoms is delayed until the late stages of development of the disease, and the most common time of presentation is the third decade of life. However, the onset of symptoms could also be delayed as late as the seventh decade. In the case reported here, the patient complained only of moderate exertional dyspnea. The SVASD congenital abnormality is thought to be due to a deficiency of the wall between the superior vena cava (SVC) and the right-sided pulmonary veins. There is volume overload on the right heart due to the left-to-right shunt through the ASD, leading to RV failure and reduced blood flow to the left ventricle. As SVASD is associated with anomalous pulmonary venous connection of some or all the pulmonary veins, this produces additional left-to-right shunting, resulting in the development of the typical symptoms[2,3].