ABSTRACT
Chest pain and dyspnoea are among the most common complaints seen in the emergency room and each symptom calls for a broad differential diagnosis. Large hiatal hernias are infrequent, but they can lead to atypical symptoms mimicking different cardiovascular, pulmonary and neoplastic diseases. We present two cases of older patients with an apparent left atrial mass on transthoracic echocardiography, which was subsequently identified as hiatal hernia by other imaging modalities. A multidisciplinary team with multimodality imaging is necessary for diagnostic work-up of chest pain and dyspnoea of non-cardiac origin and especially for a suspected mass compressing the heart, causing chest discomfort.
LEARNING POINTS
- Hiatal hernia (HH) can mimic different cardiovascular, pulmonary and neoplastic diseases.
- HH has a typical echocardiographic (2DE) presentation as an amorphous, echolucent mass with the appearance of a left atrial space-occupying lesion.
- Oral ingestion of a carbonated drink may help to distinguish between a large HH and an atrial mass by 2DE.
KEYWORDS
Hiatal hernia, left atrial mass, echocardiography; carbonated drink
INTRODUCTION
Chest pain and dyspnoea are among the most common complaints seen in the emergency room and have a considerable overlap. Each symptom calls for a broad differential diagnosis, ranging from musculoskeletal causes to potentially life-threatening emergencies, so the diagnostic work-up must be prompt. Large hiatal hernias (HH) are infrequent but can cause atypical symptoms such as chest pain and dyspnoea, and rare complications such as pulmonary oedema and even ventricular tachycardia due to the extent of the hernia and compression of the heart and pulmonary veins by organs protruding into thoracic cavity[1–3]. HH may appear as an extracardiac posterior mass encroaching on the left atrial cavity, mimicking a left atrial mass[4]. We describe two older patients with an apparent left atrial mass on transthoracic echocardiography, which was subsequently identified as HH by other imaging modalities.
CASE DESCRIPTIONS
Case 1
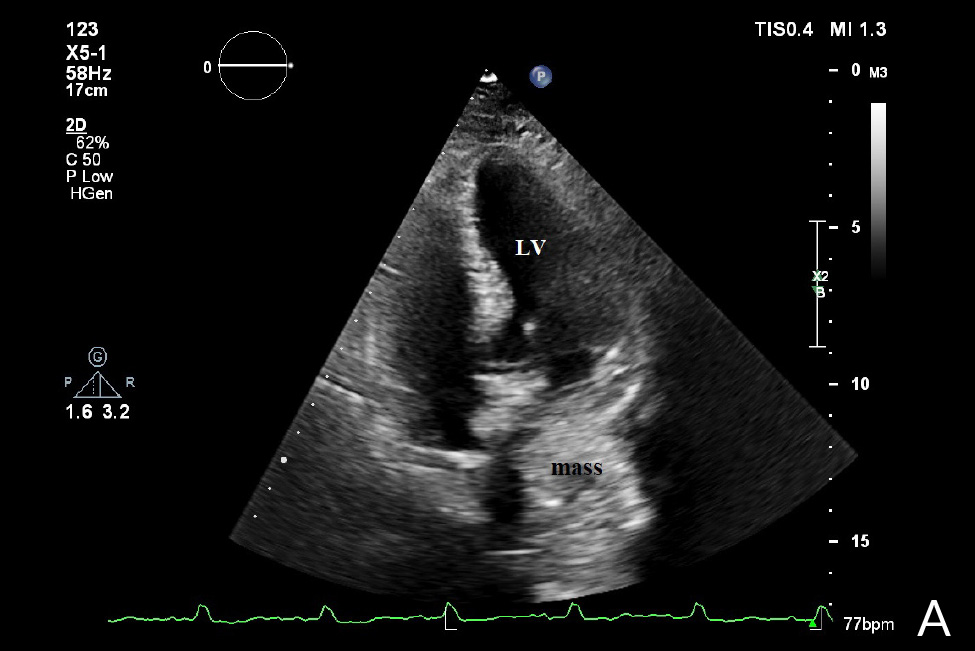
A 68-year-old woman presented with chest pain, dyspnoea and nausea for the last few hours. Her medical history was remarkable for renal failure and haemodialysis maintenance for the previous 5 years, and dialysis catheter dysfunction with subsequent replacement to the other side (the left subclavian vein) several weeks earlier. On physical examination, her blood pressure was normal (106/77 mmHg), heart rate was 81 bpm and oxygen saturation was 95% on room air. Her ECG demonstrated sinus rhythm with right bundle branch block (RBBB). The results of laboratory blood tests showed hsTnI 14.8 pg/ml (reference range: 14–18 pg/ml) and D-dimer 1872 ng/ml (normal range <500 ng/ml). A COVID-19 test was negative. The patient was admitted to the Cardiology Clinic for further investigation due to suspicion of a pulmonary embolism after replacement of the dialysis catheter. Transthoracic echocardiography demonstrated a dilated left atrium (LA), left ventricular hypertrophy with a preserved left ventricular ejection fraction (EF), and diastolic dysfunction with impaired relaxation. There was a dilated right atrium, mild tricuspid regurgitation and a normal right ventricle (RV). There were no signs of acute RV overload. Normal aorta and pulmonary arteries were present. A mass was seen on the lateral aspect of the LA which was compressing the LA. The mass was visualized in all standard and modified parasternal and apical views (Fig. 1A,B). There were no variations in transmitral flow velocities.
Figure 1. A) Apical 4 chamber view demonstrating an amorphous, echolucent mass with the appearance of a left atrial space-occupying lesion. B) Apical 3chamber view demonstrating mass on postero- lateral aspect of left atrium.
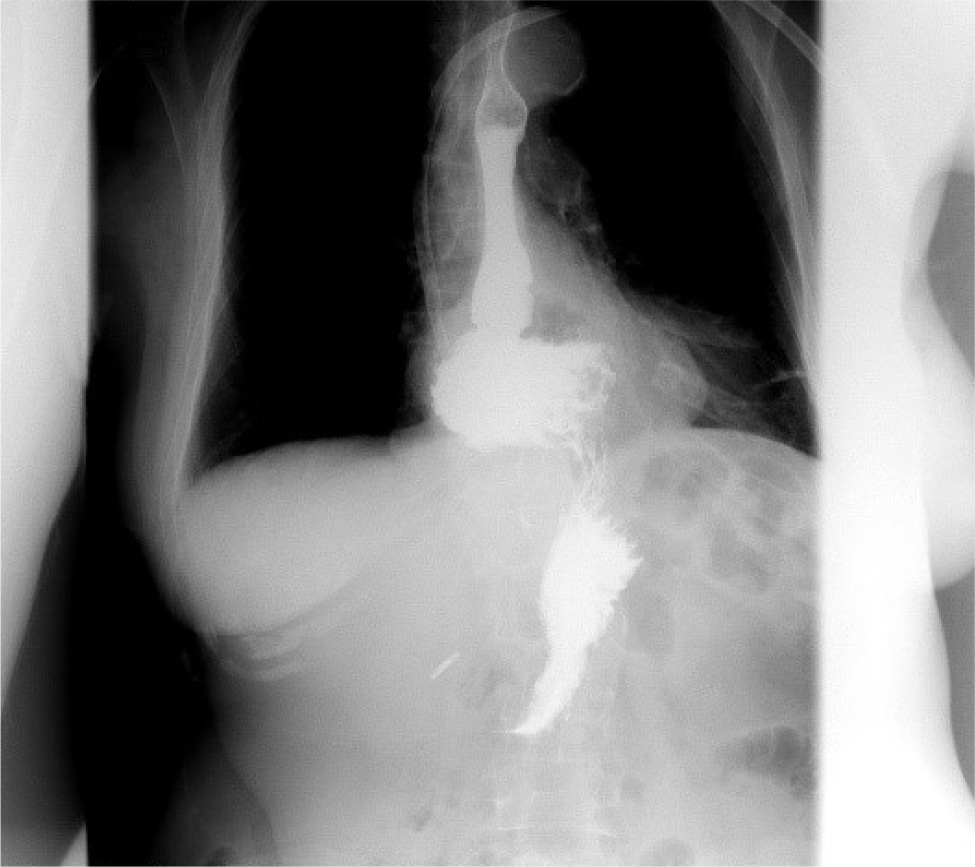
The patient was referred for upper gastrointestinal barium X-ray examination.
Intrathoracic migration of the gastroesophageal junction and a large part of the stomach above the diaphragm was confirmed (Fig. 2).
The patient underwent laparoscopic repair with complete recovery.
Figure 2. Upper gastrointestinal barium examination demonstrating intrathoracic migration of the gastroesophageal junction and large part of the stomach above the diaphragm.
Case 2
A 78-year-old woman was admitted due to chest pain with dyspnoea, pruritus and peripheral leg oedema. Her medical history was remarkable for arterial and venous thrombosis the previous year, and diabetes mellitus. A few months previously she had been admitted for right ventricular heart failure after a pulmonary embolism. Her medications included anticoagulation with apixaban, diuretics, vasodilators and an SGLT2 inhibitor. She had interrupted her medications for an undetermined time because of the pruritus. On examination, she had a normal blood pressure of 120/70 mmHg, normal heart rate of about 70 bpm, normal lung auscultation, leg oedema with reduced arterial pulses on her right lower limb, and intertriginous dermatitis. Her ECG demonstrated sinus rhythm with RBBB. Her blood test demonstrated hsTnI 12 pg/ml (reference range: 14–18 pg/ml) and D-dimer 972 ng/ml (reference range <500 ng/ml). A COVID-19 test was negative. The patient was admitted to the Cardiology Clinic for further investigation due to suspicion of a recurrent pulmonary embolism.
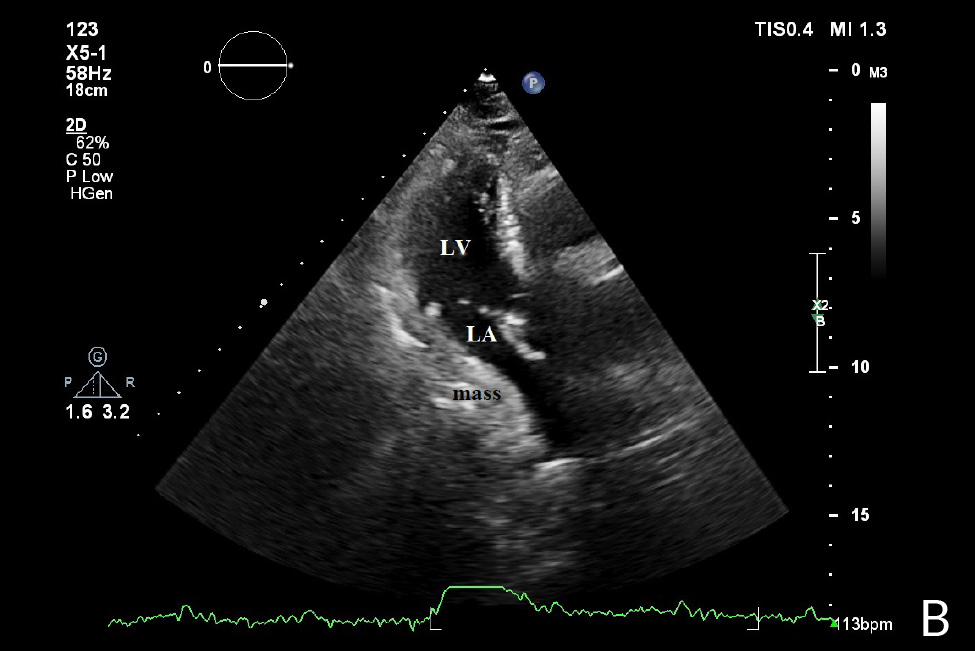
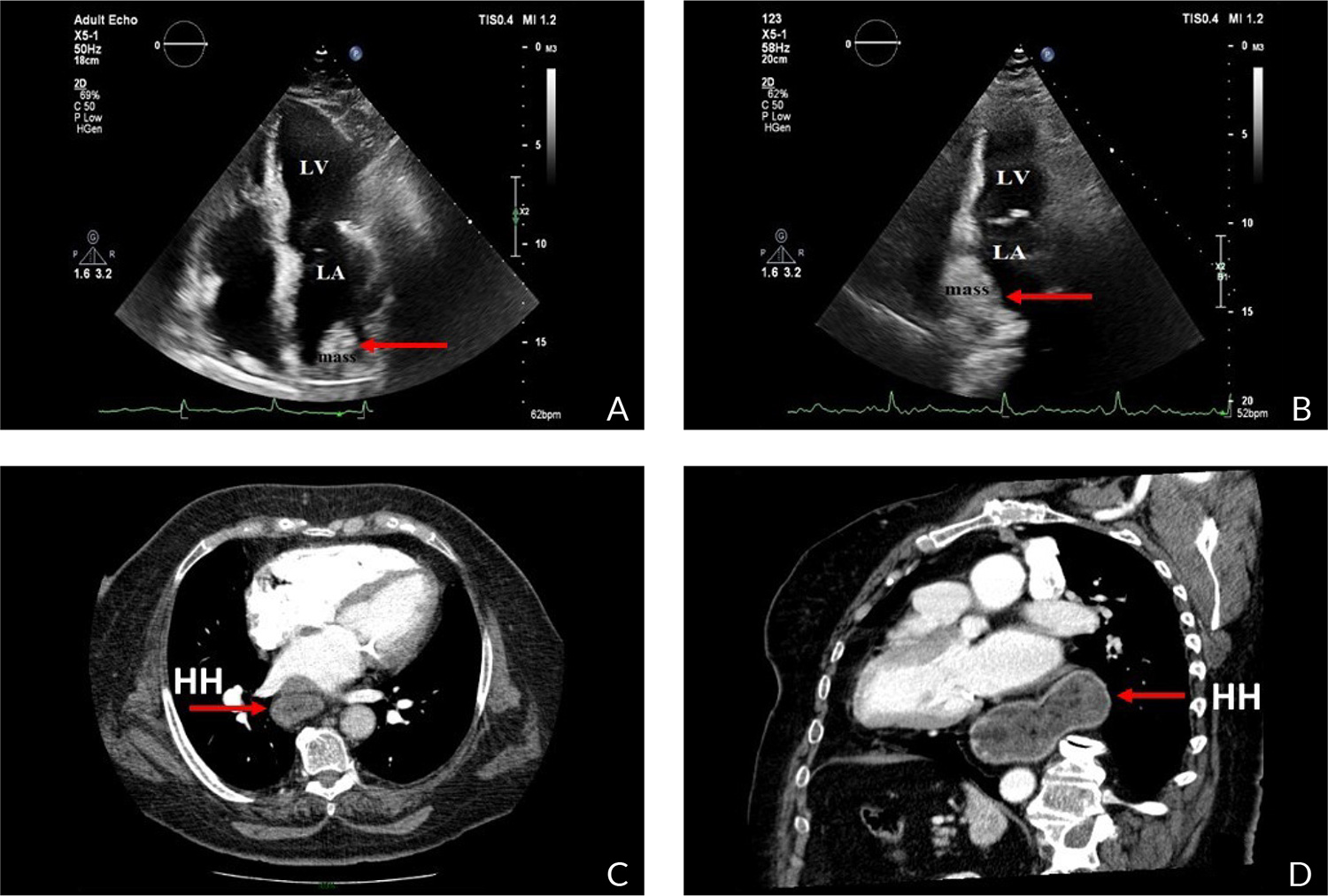
Transthoracic echocardiography demonstrated a dilated LA with an intracardial mass protruding from the right upper pulmonary vein into the LA (Fig. 3A), and another mass compressing the LA (Fig. 3B). Her left ventricle showed concentric hypertrophy with preserved EF and diastolic dysfunction with impaired relaxation. Her RV was within borderline diameters but without signs of acute RV overload. The diagnosis of left atrial thrombus originating from the right upper pulmonary vein and lung mass due to lung carcinoma or metastasis was suspected. The pruritus and rash was suspected to be a paraneoplastic syndrome.
Figure 3. A) TTE: Left atrial thrombus-like mass protruding into LA. B) TTE: Other structure resembling pulmonary neoplasia. C,D) Contrast- enhanced CT demonstrating HH compressing LA
Abbreviation: TTE- transthoracic echocardiography, LA- left atrium, LV- left ventricle, CT-computed tomography, HH- hiatal hernia
To evaluate this case, a CT scan was performed. The scan showed a large retrocardiac mass consistent with a large HH and dislocation of the stomach into the chest (Fig. 3C,D).
The patient was counselled on the need for surgery, but she refused intervention and insisted on conservative management.
DISCUSSION
These two cases of older women show that HH may mimic different cardiovascular, pulmonary and neoplastic diseases. Various intra- and extracardiac lesions can resemble the echocardiographic appearance of HH, such as left atrial thrombus and tumours, descending aorta aneurism, pulmonary neoplasia, lymphomas and non-seminomas[5].
Nishimura et al. first reported the echocardiographic visualisation of HH as an amorphous, echolucent mass with the appearance of a left atrial space-occupying lesion[6].
Subsequently, D’Cruz and Hancock[7] reported additional echocardiographic findings. They described several features that may help to distinguish between HH and atrial mass as follows: (i) HH are seen at their maximal size when the LA is visualised in a posterior plane, but are smaller or absent in more anterior planes; (ii) the degree of encroachment of the HH on the LA depends on the respiratory cycle; (iii) another typical finding is loss of the normal sharply defined sonolucency of the descending thoracic aorta due to superimposition of the HH; and (iv) most importantly, visualization of swirling echodensities following the oral ingestion of a carbonated drink[6,7].
Diagnosis of HH is typically made through an upper gastro-intestinal barium X-ray. Computed tomography and magnetic resonance imaging can be useful for further evaluation, accurately identifying the type of hernia, its contents and associated complications, and providing a roadmap for surgical planning.
CONCLUSION
In conclusion, large HH should be considered in the differential diagnosis of chest pain and dyspnoea in the emergency room. Transthoracic echocardiography is a useful diagnostic technique for the identification of intracardiac and extracardiac masses, but echocardiography images sometimes can be misleading. Multimodality imaging is necessary for suspected masses compressing the heart and causing chest discomfort.