ABSTRACT
Pulmonary arteriovenous malformations (PAVMs) are abnormal communications between the arteries and veins of the pulmonary vasculature leading to a right-to-left shunt. We report the case of a pregnant patient who presented with chest pain revealing a haemothorax secondary to the rupture of an arteriovenous malformation. The diagnosis was made during arteriography after simultaneous performance of an urgent caesarean section and thoracic drainage.
LEARNING POINTS
- The rupture of a pulmonary arteriovenous malformation in a pregnant patient is a serious and potentially life-threatening event, especially if it is not diagnosed quickly.
- It is crucial to use whatever imaging test is available whether ionizing or non-ionizing, as no thoracic imaging has been shown to adversely affect the fetus beyond embryogenesis.
- It is important for emergency doctors to consider this life-threatening condition in the differential diagnosis of atypical chest pain in pregnant women.
KEYWORDS
Pregnancy, hemothorax, pulmonary arteriovenous malformation
INTRODUCTION
Pulmonary arteriovenous malformations (PAVMs) can remain asymptomatic for a long time, but their prevalence has risen in recent years due to the increasing use of advanced imaging methods in the investigation of other diseases. Because of their wide presentation, the differential diagnosis is difficult, especially in pregnant women because of radiation protection constraints. The gold standard for diagnosis is based on non-ionizing methods, such as MRI, but this modality is not always available. Treatment of PAVMs is based on percutaneous embolization. In case of rupture, the literature considers surgery is the first treatment of choice.
CASE DESCRIPTION
A 25-year-old female patient who was 34 weeks pregnant was admitted to the emergency room with sudden onset left chest pain. The pain radiated to the left flank and back and was related to breathing. There was no history of trauma. Her medical history was relevant for supraventricular tachycardia ablated 8 years previously and hypothyroidism treated with L-thyroxine.
On admission, the patient’s heart rate was 120 beats per minute, blood pressure was 130/90 mmHg, and oxygen saturation was 95% on room air. She was afebrile. Lung sounds were absent in the left hemithorax and palpation was painful. Laboratory blood tests showed normocytic anaemia 10.1 g/dl, a predominantly neutrophilic hyperleucocytosis and increased D-dimer(2329 µg/l). Renal and thyroid function tests were within the normal ranges.
At this stage, the hypothesis of a pulmonary embolism, an aortic dissection or a pneumothorax was considered. The patient's clinical condition deteriorated rapidly 30 minutes after hospital admission. She developed severe hypovolemic shock with a systolic blood pressure of 50 mmHg accompanied by sinus tachycardia of 150 per minute. Arterial blood gas showed metabolic acidosis (pH of 6.9 and a base excess of –18).
The massive transfusion protocol was initiated and a thoracic MRI performed urgently. This demonstrated complete left pleural effusion with underlying lung collapse (Fig. 1). There was no evidence of aortic dissection. In view of these results, the diagnosis of haemothorax was raised.
After multidisciplinary discussion (surgeons, gynaecologists and intensive care doctors), the patient was taken to the operating room: an emergency caesarean section and percutaneous drainage of the pleural effusion were performed at the same time. The latter produced 800 ml of bloody fluid. Two units of blood were administered intraoperatively. There were no complications during the procedure and the baby was taken care of by the neonatal team.
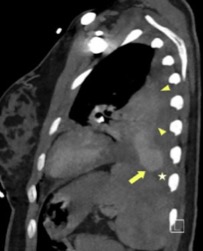
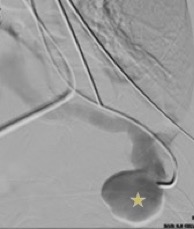
After the procedure, a thoracic CT scan was performed and demonstrated an arteriovenous malformation (AVM) in left segment 8, as well as a second centimetric nodule suspected to be a second malformation in left segment 1 (Fig. 2). The diagnosis of haemothorax due to an acute haemorrhagic complication of a PAVM within the framework of probable Osler–Weber–Rendu disease in a pregnant woman was made. The patient was then transferred for embolization of the AVM through arteriography the same day (Figs. 3 and 4). After the procedure, she was sent to the Intensive Care Unit where she remained for 72 hours before admission to a general ward.
Figure 1. MRI of the thorax (T2-weighted image, transverse axial section): left haemothorax (star) with very heterogeneous hypersignal (clot) responsible for partial atelectasis of the left lower lobe (arrow)
Figure 2. Thoracic angioscan (sagittal section): arteriovenous malformation (small star) at the pulmonary base associated with a haemothorax (large star) and partial atelectasis of the left lower lobe (arrow)
Figure 3. Pulmonary arteriography with subtraction process: selective catheterization of the left lower lobular artery with infection by contrast product: filling of the bilobulated arteriovenous malformation with contrast (star)
Figure 4. Pulmonary arteriography with subtraction process: post-embolization state without the AVM filling
DISCUSSION
PAVMs are abnormal communications between the pulmonary and systemic circulation, leading to a right-to-left shunt.
The prevalence of PAVM has been increasing in recent years, reaching about 1:2600, probably because medical imaging examinations are more readily available, especially thoracic CT scans which are widely used in lung cancer screening [1].
The clinical picture may vary from no symptoms to various and aspecific ones depending on the degree of shunt: cyanosis, dyspnoea, palpitations, heart failure and migraine [1]. AVMs can be masked by pregnancy and can lead to serious complications such as haemothorax and heart failure.
In our case the presentation of AVM was mistaken for pulmonary embolism, a more common cause of respiratory distress and chest pain.
The most common cause of PAVM is Osler–Weber–Rendu disease, an autosomal dominant genetic disorder that is defined by the presence of multiple AVMs. This condition affects 1 in 5000–10,000 people, but its incidence is probably underestimated. The second most common aetiology appears to be sporadic cases of isolated AVM, which remains a diagnosis of exclusion[1, 2].
AVMs are at high risk of rupture or bleeding during pregnancy, particularly in the last two trimesters because of the increased cardiac output and the effects of progesterone on blood vessels, with a significant lethal risk [3]. This condition is quite uncommon but should not be overlooked in the differential diagnosis of chest pain during pregnancy, which includes pulmonary or amniotic embolism, myocardial infarction, and aortic or coronary dissection [4].
Pregnant women with PAVMs are considered to be at high risk and should be closely monitored. In addition, when AVM is known to be present before pregnancy, the risk of mortality in the event of complications decreases as the clinician will be aware of the situation [5].
The management of pregnant patients is often complicated by concern that imaging examinations may harm the developing fetus [6]. However, it has been demonstrated for several decades that the radiation dose delivered by imaging modalities such as X-ray or CT scans does not exceed 50 mGy or 5 rad, which has few harmful effects on pregnancy [6–9]. However, it is of course preferable to use ultrasound and MRI when they are available and relevant.
In the case reported, the patient was able to undergo a thoracic MRI as a first-line procedure. The gold-standard treatment for AVMs has been based on percutaneous embolization for many years [1, 10, 11]. The management of AVMs discovered during pregnancy remains controversial. Embolization is widely used with good results [12–14], but it may involve a significant dose of radiation and should only be performed after the first trimester of pregnancy [13, 14].
In the case of rupture or haemorrhage in unstable patients, treatment is essentially based on surgical intervention [12, 13, 15]. With the continued technological advances in medicine, minimally invasive procedures are now possible using video-assisted thoracoscopy, offering the advantage of obtaining definitive haemostasis as well as clearing thrombi that could cause passive pulmonary atelectasis [15, 16].
In our case, the patient underwent diagnostic and therapeutic arteriography as a first-line procedure after delivery of the baby by caesarean section and after stabilization of her haemodynamic status by pleural drainage.
CONCLUSION
AVMs are probably underestimated in the population because they are frequently asymptomatic. Pregnancy increases the risk of complications, especially when the AVM is previously unknown leading to an incorrect diagnostic hypothesis. A multidisciplinary approach to the management of this pathology is essential because of the challenges associated with the choice of therapy, which must be weighed against the potential risks to the mother and fetus.