ABSTRACT
Introduction: Phenazopyridine is an over-the-counter urinary analgesic commonly used to alleviate the burning and urgency associated with lower urinary tract infections. Methaemoglobinaemia is an uncommon adverse effect of phenazopyridine use. We report a case of methaemoglobinaemia in a patient prescribed daily phenazopyridine to treat urethral and bladder irritation caused by a chronic indwelling Foley catheter.
Case description: A 55-year-old female resident of a long-term acute care facility with a chronic Foley, tracheostomy and ventilator-dependent respiratory failure was observed to have generalized dusky skin and hypoxia. Pulse oximetry was reading in the high 80s despite administration of 100% FiO2. ABG revealed paO2 of 451, oxyhaemoglobin level 75% and methaemoglobin level 22%. Medication review indicated that the patient was prescribed phenazopyridine 400 mg TID for the previous 2 months. This medication was discontinued. Considering she was chronically taking mirtazapine, she can increase risk of serotonin syndrome should she be administered first-line treatment with methylene blue. Vitamin C was thus instead administered as a second-line agent. Serial ABGs showed a rapid decline in methaemoglobin levels and an increase in oxyhaemoglobin within 2 days.
Discussion: Acquired methaemoglobinaemia is a rare adverse effect of treatment with phenazopyridine. This risk increases when drug dosage and duration exceed manufacturer specifications. Treatment typically includes cessation of the offending drug and administration of methylene blue in severe cases. A thorough medication reconciliation should be performed prior to methylene blue initiation, as patients taking serotonergic medications (for example, MAOIs, SSRIs, SNRIs, TCAs) are at increased risk of serotonin toxicity with co-administration of methylene blue. In these instances, ascorbic acid/vitamin C can be chosen as an alternative treatment agent.
Conclusion: Work-up of refractory hypoxia should involve a thorough review of medications as even some over-the-counter drugs can cause the fatal side effect of methaemoglobinaemia. Treatment with vitamin C should be considered over methylene blue if serotonergic medications have been recently prescribed in order to avoid risk of serotonin syndrome.
LEARNING POINTS
- Methaemoglobinaemia is an uncommon, life-threatening adverse effect of phenazopyridine use. Presentation depends on the severity of the disorder, ranging from headache, weakness, lightheadedness and dyspnoea, to arrhythmias, confusion, seizures and multiorgan failure.
- Workup of refractory hypoxia should involve a comprehensive medication review as even some over-the-counter drugs can cause methaemoglobinaemia.
- Management of methaemoglobinaemia involves cessation of the offending drug, administration of supplemental oxygen and treatment with methylene blue (1–2 mg/kg) if MetHb >30%, or for symptomatic patients with MetHb >20%. Vitamin C can be used as an alternative agent if there is a contraindication to methylene blue (for example, with patients simultaneously receiving serotonergic medications and/or those with G6PD deficiency).
KEYWORDS
Methaemoglobinaemia, phenazopyridine, urinary tract infection, serotonin syndrome, arterial blood gas, methylene blue, case report
INTRODUCTION
Phenazopyridine (Pyridium) is a urinary analgesic available both over-the-counter and as a prescription, commonly used to alleviate the burning, urgency and discomfort associated with irritation of the lower urinary tract mucosa in the setting of lower urinary tract infections. Reported side effects of its use include headaches, vertigo, rash, pruritus, gastrointestinal (GI) disturbances, skin pigmentation, orange discolouration of body fluids including tears and urine, haemolytic anaemia in G6PD-deficient patients with chronic overdose and transient acute renal failure [1]. Serious reactions include anaphylaxis/anaphylactoid reactions and methaemoglobinaemia.
Methaemoglobinaemia arises when the iron component of heme oxidizes to the ferric form (Fe3+) rather than the more soluble ferrous form (Fe2+). Because ferric iron is unable to bind oxygen, it cannot be delivered to the tissues, resulting in hypoxia. Methaemoglobinaemia may be secondary to genetic defects in red blood cell metabolism or haemoglobin structure, or it may be acquired following exposure to oxidant drugs and toxins [2].
Methaemoglobinaemia is an uncommon adverse effect of phenazopyridine use, with fewer than 50 documented cases since 1951 identified in our review of the literature. We report a case of methaemoglobinaemia in a patient prescribed daily phenazopyridine to treat urethral and bladder irritation caused by chronic indwelling Foley catheter use for urinary retention.
CASE DESCRIPTION
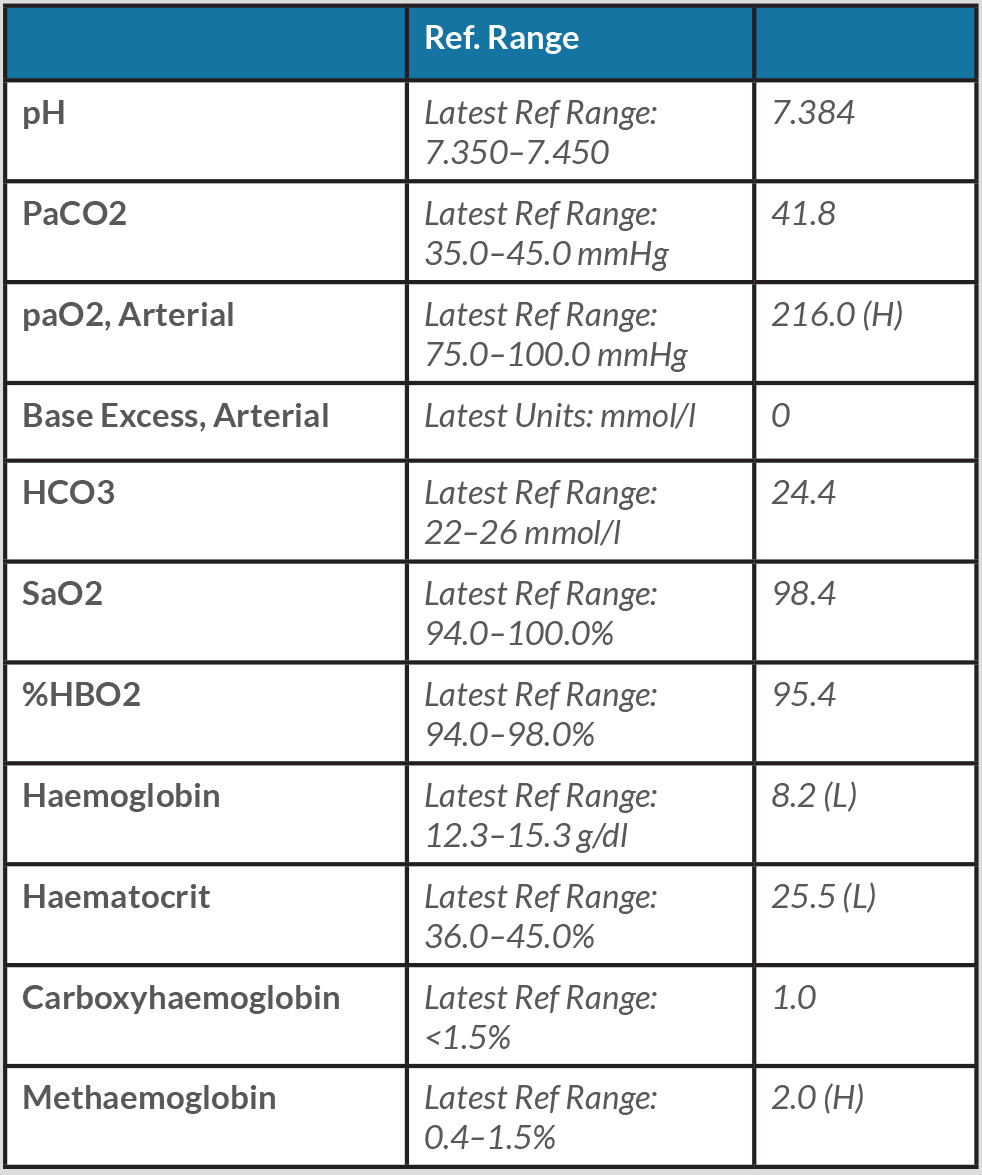
A 55-year-old female was transferred to the hospital from a long-term acute care facility due to cyanotic skin appearance and persistently low oxygen saturation as reported by finger pulse oximetry. The patient had a history of ventilator-dependent chronic respiratory failure s/p tracheostomy and PEG tube, avascular necrosis of bilateral hips, urinary retention with chronic indwelling Foley catheter, recurrent UTIs, heart failure with reduced ejection fraction (EF 20%), chronic macrocytic anaemia and pulmonary emboli (PE). Despite increasing FiO2 to 100% on the ventilator, the patient remained with oxygen saturation in the high 80s. She was normotensive and afebrile. She was alert and oriented to person, place and time. Initial ABG was significant for pH of 7.40, pCO2 40.3, paO2 of 451, oxyhaemoglobin saturation 75% and methaemoglobin level of 22% at her facility. Follow up ABGs after several hours on 100% FiO2 revealed the results shown in Table 1.
Chronic macrocytic anaemia Hgb 7.0 and MCV 104 were noted, but otherwise the laboratory results were unremarkable. In light of elevated methaemoglobin levels, the patient’s medication list was carefully reviewed. She had been prescribed lorazepam as needed for anxiety, mirtazapine and phenazopyridine. The patient indicated that she was taking phenazopyridine 400 mg 3 times a day for the past 2 months for treatment of “urinary tract spasms”. Phenazopyridine was stopped, and the patient was managed with ventilator support, high FiO2, and vitamin C 1,000 mg IV 3 times a day for 2 days. Methylene blue was avoided in this patient considering her chronic use of mirtazapine and the heightened risk of developing serotonin syndrome. Repeat ABG 2 days later showed resolution of methaemoglobinaemia and the results are given in Table 2.
Her cyanosis resolved. She was eventually discharged back to her long-term acute care facility in a stable state.
Table 1. ABG results after several hours on 100% FiO2
Table 2. ABG results after treatment showing resolution of methaemoglobinaemia
DISCUSSION
Phenazopyridine is an azo dye used to treat dysuria for symptomatic relief of pain, urgency, burning, frequency and other discomforts arising from irritation of the lower urinary tract mucosa. It has a recommended adult dose of 100 to 199 mg given 3 times a day, with a duration limited to 2 days. Methaemoglobinaemia can be caused by a genetic flaw in haemoglobin structure or red blood cell metabolism (haemoglobin M disease or cytochrome b5 reductase deficiency), or it can be acquired after exposure to oxidant medications or toxins, as in the present case. Acquired methaemoglobinaemia is seen in <1% of patients treated with phenazopyridine, but this risk is increased when drug dosage and duration exceed manufacturer specifications. Typically, methaemoglobin is converted back to haemoglobin by an enzymatic process. In the case of phenazopyridine poisoning, this cascade is circumvented, resulting in methaemoglobin build-up. Other medications that have been associated with acquired methaemoglobinaemia are local anesthetic agents and dapsone [3].
Presentation of methaemoglobinaemia depends on the severity of the disorder, ranging from headache, weakness and lightheadedness, to dyspnoea, confusion and seizures. Levels >50% can cause death. Co-oximetry is the test of choice for diagnosis. Treatment includes cessation of the offending drug and administration of methylene blue (1–2 mg/kg) if MetHb >30%, or for symptomatic patients with MetHb >20%. Vitamin C is used as an alternative to methylene blue, which can precipitate haemolysis in G6PD-deficient patients. Hyperbaric oxygen and exchange transfusions are reserved for refractory cases [4].
Our patient was treated with vitamin C instead of methylene blue as we were concerned about the possible sequela of serotonin syndrome with concomitant use of mirtazapine. Methylene blue is known to interact with serotonergic psychiatric medications and can cause serious CNS toxicity. Per FDA guidelines, if methylene blue is indicated for a patient taking a serotonergic drug, the serotonergic drug must be stopped immediately, and the patient should be monitored closely for 2 weeks (5 weeks if fluoxetine was taken), or until the last dose of methylene blue is given, whichever comes first. Twenty-four hours following the last dose of methylene blue, treatment with the serotonergic psychiatric medicine may be restarted [5]. In the case described above, our patient was treated with vitamin C. Skin pallor resolved and over the course of 2 days, serial ABGs revealed a significant reduction in methaemoglobin as well as a rapid rise in oxyhaemoglobin, when the patient was pronounced stable.
CONCLUSION
Methaemoglobinaemia is a life-threatening condition that must be diagnosed quickly. Meticulous investigation of refractory hypoxia in acute settings should involve a thorough review of the patient’s medication history as methaemoglobinaemia is a rare but potentially fatal side effect of several common medications, including those purchased over the counter (such as phenazopyridine).