ABSTRACT
We report the case of a 61-year-old man admitted to our emergency department with fever. At admission, he was hypotensive and tachycardic. In the initial investigation, elevation of inflammatory parameters, acute kidney injury (Kidney Disease Improving Global Outcomes (KDIGO) 3), hyperbilirubinemia, and hepatic cytocholestasis were evident. Empirical antibiotic therapy was started, after sepsis was assumed without an identifiable cause. His condition took an unfavorable clinical course, with respiratory failure, hepatosplenomegaly, pancytopenia, hyperferritinemia and hypofibrinogenemia. Microbial culture studies and a general immunological study were negative and lymphoproliferative disease was therefore excluded. Bone marrow aspirate revealed hemophagocytosis without granulomas. A diagnosis of hemophagocytic lymphohistiocytosis was assumed and pulse methylprednisolone therapy initiated. As this resulted in only a transient improvement, immunoglobulin and rituximab were initiated as a second-line therapy. The patient sadly had an unfavorable outcome despite all measures undertaken. In the postmortem study, Mycobacterium tuberculosis complex was isolated in the bone marrow aspirate, which led to the postmortem diagnosis of disseminated tuberculosis and angioinvasive pulmonary aspergillosis. The clinical presentation of disseminated tuberculosis is non-specific and hemophagocytic lymphohistiocytosis is one of its rare presentations. The mortality rate of hemophagocytic lymphohistiocytosis is high and increases with delayed diagnosis of the underlying condition and respective treatment.
LEARNING POINTS
- Hemophagocytic lymphohistiocytosis should be considered in patients presenting with fever, lymphadenopathy, splenomegaly, cytopenias, hyperferritinaemia and hypertriglyceridemia.
- Despite its rarity, tuberculosis should be considered as an etiology of hemophagocytic lymphohistiocytosis and, if suspected, antituberculosis therapy should be initiated early, even in the absence of a definite diagnosis.
- Immunosuppressant therapy increases the risk of opportunistic infections, which establishes the need for prophylactic antibiotic, antifungal, and antiviral drugs.
KEYWORDS
Hemophagocytic lymphohistiocytosis, disseminated tuberculosis, immunosuppressive therapy, aspergillosis, intensive care
CASE DESCRIPTION
We report the case of a 61-year-old man, a former smoker with a history of chronic alcoholism, chronic lung disease, and pleuropulmonary tuberculosis 15 years prior to presentation. He was admitted to the emergency department (ED) in our hospital due to fever with rigors and a 2-day history of myalgia. He also reported pollakiuria during the previous months. He had no respiratory symptoms. In the ED, there was no impairment of mental function, he was eupneic with normal pulmonary auscultation, peripheral perfusion was adequate, and abdominal palpation was normal. Hypotension and tachycardia were evident (blood pressure 80/40 mmHg; heart rate 120 bpm), with rhythmic heart sounds. Initial blood analysis revealed elevated C-reactive protein (196 mg/L), acute kidney failure (creatinine 3.37 mg/dL; urea 76 mg/dL) and elevated liver enzymes (alanine aminotransferase 235 U/L; aspartate aminotransferase 410 U/L; gamma-glutamyl transferase 375 U/L; alkaline phosphatase 158 U/L) and total bilirubin of 2.39 mg/dL. The remaining blood analysis was normal, including hemoglobin (15.9 g/dL), white blood cells (5.170/uL) and platelets (283,000/uL). Urinalysis was normal as well as simple chest radiography and transthoracic echocardiography. An abdominal ultrasound scan revealed hepatosplenomegaly but no signs of urinary obstruction. A diagnosis of sepsis was assumed without an identifiable origin. Fluid therapy and empirical treatment with ceftriaxone and doxycycline were started, with favorable progression, including hemodynamic stabilization, apyrexia, and renal failure improvement. The patient was then admitted to a medical ward for treatment and diagnostic work-up.
Subsequently, progressive deterioration of his mental status became evident, initially with occasional visual hallucinations followed by hetero-aggressive behavior and persistent agitation. At first, it was assumed to be delirium tremens, refractory to benzodiazepines and antipsychotics, which led to his admission to a high dependency unit (HDU) and dexmedetomidine commenced.
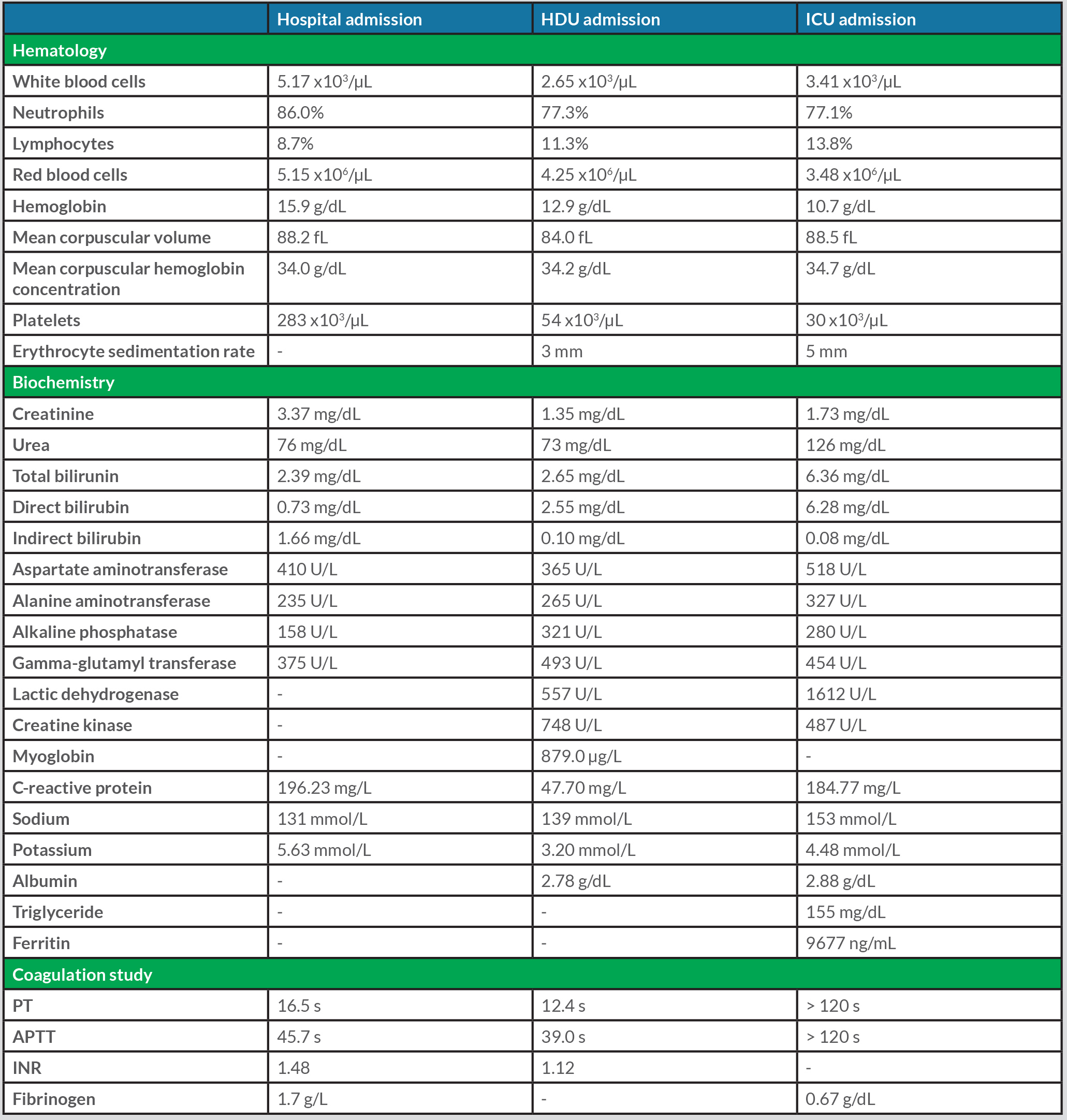
During his HDU stay, his clinical condition and blood analysis worsened. His mental status continued to deteriorate, with progressively decreasing levels of consciousness. Computerized tomography (CT) of the brain was normal as were cerebral brain fluid and ammonia levels. The patient’s fever recurred, with an elevated procalcitonin level, for which the antibiotic therapy was upscaled to piperacillin/tazobactam and later to meropenem and vancomycin. Blood analysis (Table 1) revealed progressive normocytic normochromic anemia, thrombocytopenia, leucopenia with lymphopenia, elevated ferritin and lactic dehydrogenase but with normal levels of triglycerides. Liver failure was also evident, with severe coagulopathy and hypofibrinogenemia. An exhaustive microbiological study was performed, as presented in Table 2. Thoracic, abdominal and pelvic CT revealed sparse areas of ground-glass lesions, small bilateral lung effusions, hepatosplenomegaly and splenic infarction. However, transthoracic and transesophageal echocardiography were normal, revealing no sign of infectious endocarditis. Progressive respiratory failure was evident, which required intubation, invasive mechanical ventilation, and admission to the intensive care unit (ICU).
Given the fever, hepatosplenomegaly, progressive pancytopenia, raised ferritin and hypofibrinogenemia, the patient presented with five of the eight second-line criteria required for the diagnosis of hemophagocytic lymphohistiocytosis (HL). A bone marrow biopsy was therefore performed, demonstrating hemophagocytosis with no evidence of granulomas, which confirmed the diagnosis of HL. The screening for acquired causes of HL is presented in Table 2. Without clear etiology for HL, pulse methylprednisolone therapy for 4 days was started. There was only a transient decrease in ferritin levels observed, without clinical improvement, which prompted the administration of immunoglobulin for 5 days and a single dose of rituximab. An unfavorable outcome ensued, with the patient experiencing severe acute respiratory distress syndrome (ARDS), refractory shock, and anuric acute kidney injury, sadly leading to his death on day 7 of his ICU stay.
Postmortem microbiology of the bone marrow aspirate revealed Mycobacterium tuberculosis complex, resulting in a postmortem diagnosis of disseminated tuberculosis, with liver and bone involvement and angioinvasive pulmonary aspergillosis.
Table 1.Laboratory data at hospital admission, High Dependency Unit (HDU) admission and Intensive Care (ICU) admission
Table 2. Screening for acquired forms of hemophagocytic lymphohistiocytosis
DISCUSSION
While tuberculosis often presents as a subacute infection, it may on rare occasions manifest as sepsis or septic shock, conferring a poor outcome with a hospital mortality rate of 80%[1]. The risk factors for tuberculosis at an individual level are immunosuppressive conditions (namely HIV co-infection), malnutrition, young age, diabetes, socioeconomic factors, smoking habits, alcohol consumption, and indoor air pollution[2]. The clinical presentation of sepsis secondary to tuberculosis includes hematologic abnormalities such as disseminated intravascular coagulation, altered mental status, renal insufficiency, refractory hypotension, and ARDS, but it may be indistinguishable from bacterial sepsis[1,3]. The majority of cases present with an abnormal chest radiography pattern, usually with a diffuse multilobar consolidation[4].
Risk factors or characteristics of tuberculosis septic shock are, however, insufficiently specific to allow differentiation from other causes of septic shock[4], which could explain the lack of suspicion of tuberculosis with our patient. Furthermore, despite presenting with individual risk factors for tuberculosis, such as alcoholism and being a former smoker, and the clinical features of tuberculosis sepsis, such as hematologic abnormalities, altered mental status, renal failure, refractory hypotension and ARDS, our patient did not present with indicative radiological features. His sputum for the acid-fast stain was negative and non-caseating granulomas were not found in the bone marrow biopsy. Due to this lack of suspicion, antituberculosis therapy was not started.
Despite our patient having initially presented with sepsis, his condition later evolved to fulfilling six of eight diagnostic criteria for HL, precipitating an early aggressive screening for the etiology of the underlying disease. HL is a hyperinflammatory hyperferritinemic immune response driven by T cells, associated with a potentially fatal cytokine storm[5]. It is classified as primary which is most common in childhood and secondary which is mainly seen in adulthood. It can also be secondary to a severe infection, most commonly of viral etiology, but it has also been associated with fungal, parasitic, and bacterial infections, including Mycobacterium tuberculosis, autoimmune diseases, and malignant tumors[5–7].
According to 2004 guidelines for hemophagocytic lymphohistiocytosis, the diagnostic criteria of HL is established if molecular diagnosis is consistent with HL and/or when five of the eight diagnostic criteria for HL are fulfilled[8]. Once a diagnosis of HL is established, the underlying condition must be investigated, as its treatment is based on treating the underlying pathology as well as supportive care and immunosuppressant therapy, including corticosteroids and/or etoposide to prevent the life-threatening cytokine storm[5,7,8]. Timely treatment of the underlying condition improves the outcome of life-threatening HL[5,8].
HL secondary to disseminated tuberculosis is rare and reported in about 4.8–16.7% of patients with infection-associated HL. It is a potentially life-threatening disorder with a high ICU admission rate (71%) and mortality rate (38%), with 86% of cases occurring in immunocompromised individuals[6]. The immunosuppressants administered to suppress the overactivation of the immune system increase the risk of opportunistic infections, giving rise to prophylactic antibiotic, antifungal, and antiviral therapy[5]. The postmortem of our patient also revealed angioinvasive pulmonary aspergillosis, reflecting the high level of induced immunosuppression in this patient. Our case report highlights the importance of an early diagnosis of the underlying disease of HL and the importance of considering tuberculosis as an infective cause. Finally, a low threshold for initiating empirical antituberculosis therapy must be held.