ABSTRACT
Coronary artery fistulae (CAF) are a rare entity describing abnormal communication between a coronary artery and cardiac chamber or a large intrathoracic vessel and are seldom described in the literature[1]. These fistulae can be either congenital or acquired. Often incidental in finding, CAFs can have serious clinical sequelae, and should be duly reported and discussed with the cardiac team. More than 50% of patients with CAFs may be asymptomatic; 34% may report chest pain; 13% may have symptoms of heart failure, and a minority of 2% may suffer from endocarditis and arrhythmias[2]. The largest series to our knowledge was reported by the Cleveland Clinic, which found 225 patients with incidental CAF out of 126,595 coronary catheterizations (incidence of 0.18%), performed during a span of 28 years[3].
Multiple CAFs are an even rarer entity and only a handful of cases have been reported in the literature to date. Few cases of double CAFs have been reported that describe two different feeder coronary arteries giving rise to separate drainage sites[3]. In our report, however, we describe a new entity: a single-feeder coronary vessel communicated with two drainage sites. Our case is curiously unique, in which we report a single artery, originating from the right coronary artery (RCA) with double drainage sites – one to the left pulmonary artery and the second to the left bronchial artery.
LEARNING POINTS
- Cardiac CTA confers added value in identification of both normal and abberant coronary vessels.
- Maximal Intensity Projection (MIP) & 3D volumetric reconstructions are of added value in every cardiac CTA due to its all-encompassing view of the heart. These techniques have been incorporated and are routinely used in all our Cardiac CTAs.
- Curved MIP reconstructions of the coronary arteries, combined with 3D volumetric reconstruction of the heart, allows comprehensive evaluation of the coronary arteries, especially in complicated cases.
- Due to its anatomic and 3-dimensional properties – not only of the heart – but of the entire thorax, cardiac CTA is ideal for evaluation of fistulae between coronary arteries and adjacent vessels.
- Cardiac CTA may facilitate the illustration of single-feeder, double-drainer coronary artery fistulae, which was not clearly identified in our case during conventional coronary angiography.
KEYWORDS
Coronary artery fistula, abberant coronary artery, cardiac CT, cardiac catheterization
CASE PRESENTATION
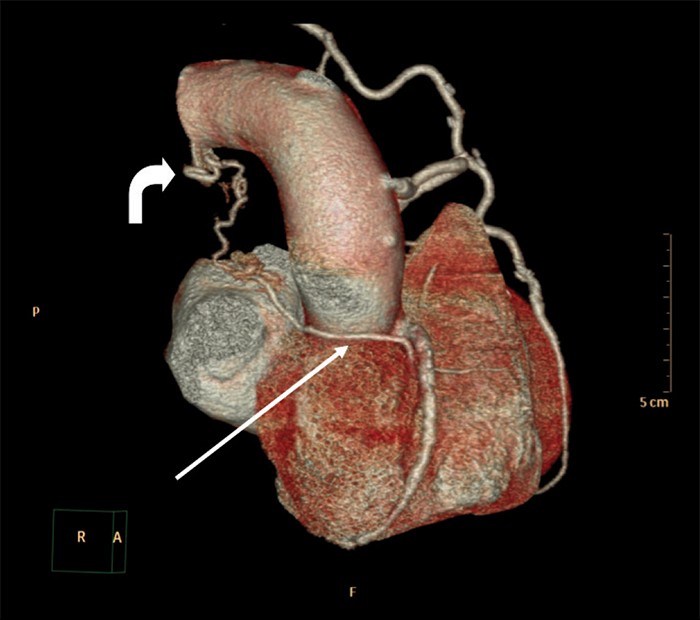
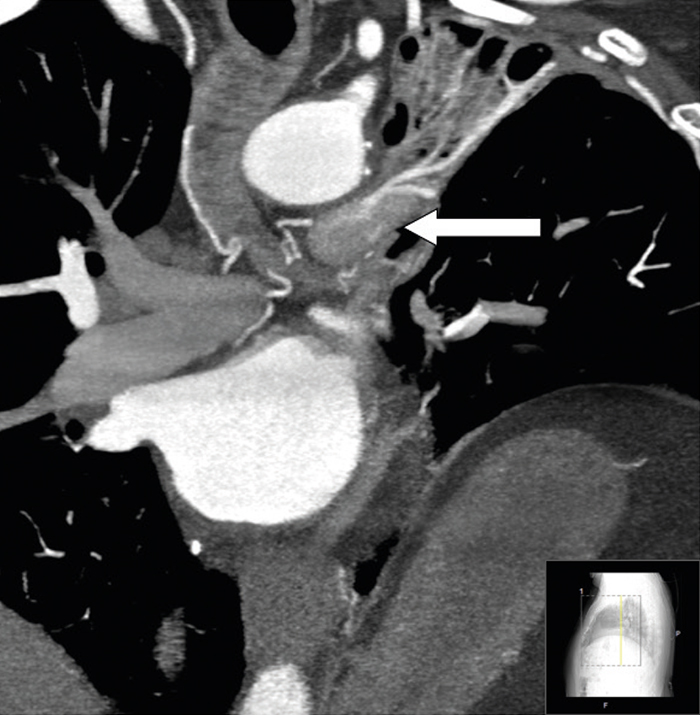
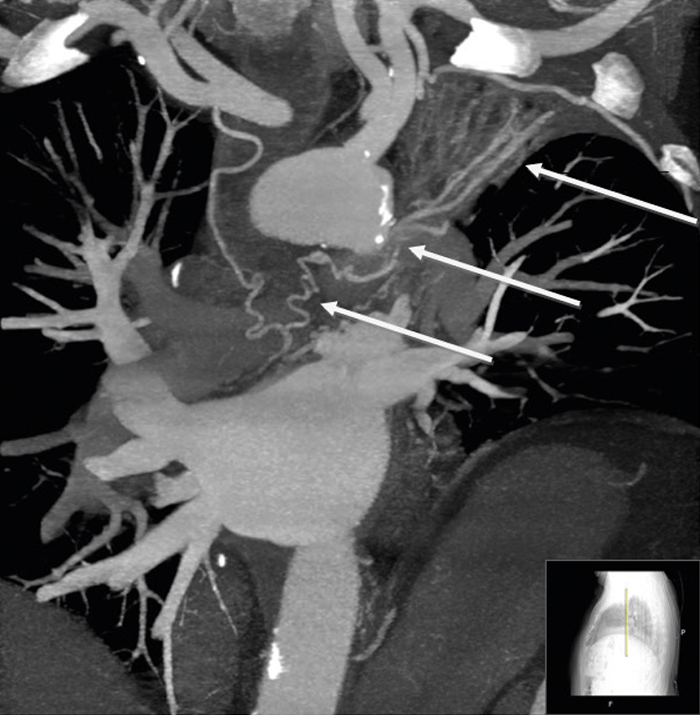
A 65-year-old male with longstanding metabolic syndrome and ischemic heart disease presented to the ED with chest pain and dyspnea. Fifteen years earlier, the patient underwent coronary artery bypass, which included Left Internal Mammary Artery to Left Anterior Descending Artery (LIMA-to-LAD) and SVG-to-M1. Due to his clinical manifestation, a diagnostic angiography study was performed, and a stent was inserted into a tight stenosis of the proximal SVG. Incidentally, a tortuous vessel was revealed: it originated from the RCA, passed upward and drained into a large vessel, where a turbulent steal was noted at this site. It looked like it could be the pulmonary artery, however on cardiac CT this tortuous vessel was found to be a branch of the RCA to the right atrium (Fig. 1). This branch was relatively large in caliber and passed to the right of the ascending aorta, communicating with the upper border of the left main pulmonary artery (left to right shunt). At this site, a vague enhancement was noticed (407 HU) compared to the main pulmonary artery and right pulmonary artery (157 HU) (Fig. 2). Furthermore, this vessel continued north-west and passed through the posterior mediastinum and to the left. A communication was found with a conglomerate of mediastinal vessels, which supplied an atelectatic left-upper-lobe containing multiple bronchiectases (left to left shunt), (Fig. 3). These atelactases and bronchiectases of the left lung were well known from previous chest x-rays, at least four years prior.
Figure 1: 3D reconstruction demonstrating an aberrant coronary vessel originating from a right coronary artery (RCA) branch (white arrow) and its tortuous course posterior to the ascending aorta (curved arrow).
Figure 2: Maximum intensity projection (MIP) Coronal Section. Note the slight attenuation difference at the site of the RCA-LTPA fistula (block arrow).
Figure 3: Maximum intensity projection (MIP) Coronal Section, demonstrating the tortuous course of the RCA vessel and its connection to the interlay of dilated bronchial vessels (white arrows).
DISCUSSION
CAFs can be classified into two categories: congenital and acquired, with 64% of reported CAFs thought to be congenital. Acquired CAFs have been reported as a complication of deceleration accidents, percutaneous transluminal coronary angioplasty (PTCA), repeated endomyocardial biopsies in heart transplant recipients, permanent ventricular pacing leads, or cardiac surgery[2,3]. Since this CAF was not demonstrated in previous studies and was not directly visualized upon sternotomy, we postulate that this double CAF is likely the product of an iatrogenic cause. Our patient underwent coronary artery bypass surgery (CABG) fifteen years prior, which is a well known cause of CAF creation[4]. It has been posited by several authors describing the handful of multiple CAFs that as cardiac imaging, specifically cardiac computed tomography (CT) scans, become more prevalent, more CAFs, which were previously unknown, will become apparent [5].
The etiology of the single-feeder double-drainer CAF observed in our patient may be due to chronic bronchiectasis and atelectasis of the left upper lobe (LUL). Several reports have described this peculiar correlation between bronchiectasis and CAF[5]. This LUL pathology likely caused increased bronchial artery demand, and according to the sparse literature, likely created the distal fistula drainer. Since the feeder artery coursed in close proximity to the left pulmonary artery, the proximal fistula drainer at the pulmonary artery could have been formed by sheer proximity.
Interestingly, the pulmonary artery has been reported as the most prevalent site of CAF drainage (68%) out of all drainer-vessels[1]. It can be hypothesized that separate entities created the separate drainers or that the pulmonary artery fistula arose first. However the relevance of this "chicken-egg" quandary is quite irrelevant in our case, as the importance of finding a single salient explanation for this curious single-feeder double-drainer CAF is slight.
The clinical issue, however, concerning this patient’s condition was whether or not to attempt to surgically correct this CAF. Practically, this CAF may be treated by insertion of cover-stent in the proximal RCA, occluding the orifice of this feeder. Similar procedures have been reported by ligation of the orifice of the feeder[6]. Kamiya et al. report a 98% morbidity-free ten-year survival rate of those who underwent surgical correction, with a low-to-nil re-op rate, most likely due to initial low flow through the fistulae[1]. However, in our case, we had to account for both drainage sites being shut off after occluding the single feeder. After deliberation, we decided to assess the hemodynamic significance of the fistula, as the literature supports that large, proximal and hemodynamically significant fistulas should be closed, either by surgery or by catheter techniques[6].
Spirometry results were within normal limits. Bronchoscopy yielded normal findings, except for an aberrant bronchus to the lingula. While the patient failed to consent for invasive evaluation of the shunt, a non-invasive assessment was performed by transthoracic echocardiography (TTE). TTE showed normal chamber sizes, preserved global left and right ventricle function, and no significant valvular pathology. There was mild tricuspid regurgitation with low calculated systolic gradient, conforming normal estimated pulmonary artery pressure. The pulmonary trunk was only mildly dilated at 25 mm. Shunt estimation was performed by a volumetric flow calculation of left and right ventricle stroke volumes, by multiplying outflow tract velocity time integral (VTI) by calculated outflow tract cross-sectional area [LVOTd 2.0 cm; LVOT VTI 21.3 cm; RVOTd 3.0 cm; RVOT VTI 13 cm]. Qp/Qs of 1.4 was measured, thus there was no evidence for significant volume overload.
In addition to the absence of demonstrable hemodynamic significance of this curious CAF, so too, after PTCA of the saphenous vein grafts (SVG) and stent insertion, the patient's chest pain and dyspnea subsided. Most probably, this CAF although containing a left-to-right shunt and left-to-left shunt to the LUL, does not represent a large enough cardiac blood supply, or "steal" to cause clinical symptoms. If left untreated, however, this CAF could potentially cause left heart failure in the future due to increasing steal of coronary blood supply in an aging heart. Furthermore, right heart failure could arise in the future as well, to due volume overload through the pulmonary artery (left-to-right shunting). At this point in time, without evidence of volume overload and in the absence of chest pain, a conservative approach was chosen. Upon review of the literature, a large number of CAFs have been treated conservatively, with even a small number of fistulae closing spontaneously. Most fistulas may be successfully treated conservatively – up to 78% one report cited[2], especially those with small shunts of Qp/Qs <1.5.
CONCLUSION
While most CAFs tend to originate from the left, cases have been reported of right coronary artery origin. Likewise, the most prevalent drainage site has been reported as the pulmonary artery (68%) with other destinations being the coronary sinus, right atrium, right ventricle, and very rarely the bronchial artery[1]. Our case is an unusual presentation of a patient with a coronary fistula originating from a proximal stenotic RCA and connecting to the left branch of the pulmonary artery and to an aberrant bronchial artery associated with left upper lung bronchiectasis, creating both left-to-right and left-to-left and shunts.
Without a prior single-feeder, double drainer CAF being described in the literature, our decision to treat conservatively was based upon the previously described single-feeder, single-drainer CAFs being treated conservatively with success, combined with the overall clinical picture, indicating a prudent "stay and wait" approach. Since cardiac computed tomography angiograms (CTA) are increasingly being performed, incidental CAF findings will likely appear more often, and similar questions as to operate or to treat conservatively may arise. As noted in our case, cardiac CTAs may become the modality of choice for CAF evaluation due to its spatial resolutions and volumetric 3D reconstructions. It may be posited that previously thought of single-feeder, single-drainer CAFs by angiography might actually be discovered to be demonstrated quite differently and considered a separate and peculiar entity, as described in our case.