ABSTRACT
Objectives: Agents targeting the epidermal growth factor receptor (EGFR)-mediated signalling pathway are increasingly being used for the treatment of advanced lung, pancreatic, colorectal and head and neck cancers.
Case presentation: Here, we report the first case of eruptive seborrhoeic keratosis following panitumumab treatment, an anti-EGFR monoclonal antibody, in a 73-year-old patient with stage 4 (IV) colorectal cancer with hepatic metastasis.
Conclusion: While panitumumab is an emerging therapy for RAS wild-type metastatic colorectal cancer, physicians should consider panitumumab as a potential cause of eruptive seborrhoeic keratosis.
LEARNING POINTS
- Panitumumab is a fully human monoclonal antibody targeting the epidermal growth factor receptor (EGFR), indicated for the treatment of wild-type RAS metastatic colorectal cancer.
- Dermatologic toxicity of all grades occurs in more than 90% of patients treated with EGFR inhibitors.
- To the best of our knowledge, we have reported here a rare side effect; panitumumab-induced eruptive seborrhoeic keratosis.
KEYWORDS
Panitumumab, eruptive seborrhoeic keratosis, drug-induced eruption
INTRODUCTION
Panitumumab is a fully human monoclonal antibody targeting the epidermal growth factor receptor (EGFR), indicated for the treatment of wild-type RAS metastatic colorectal cancer, either alone or combined with chemotherapy[1]. Dermatologic toxicity of all grades occurs in more than 90% of patients[1]. To the best of our knowledge, panitumumab-induced eruptive seborrhoeic keratosis has not been previously reported. Here, we report the first case of panitumumab-induced eruptive seborrhoeic keratosis in a 73-year-old patient with stage 4 (IV) colorectal cancer with hepatic metastasis.
CASE DESCRIPTION
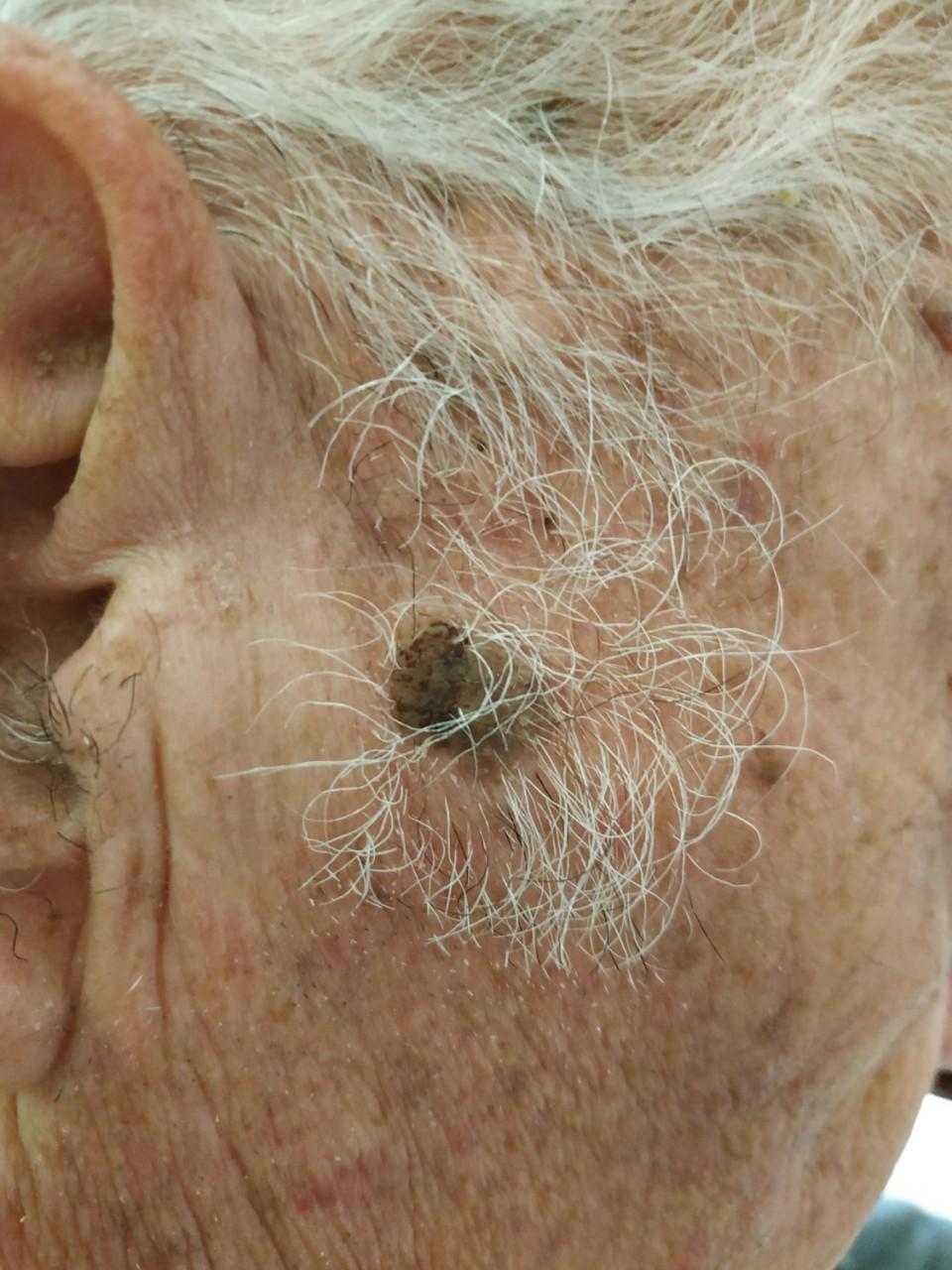
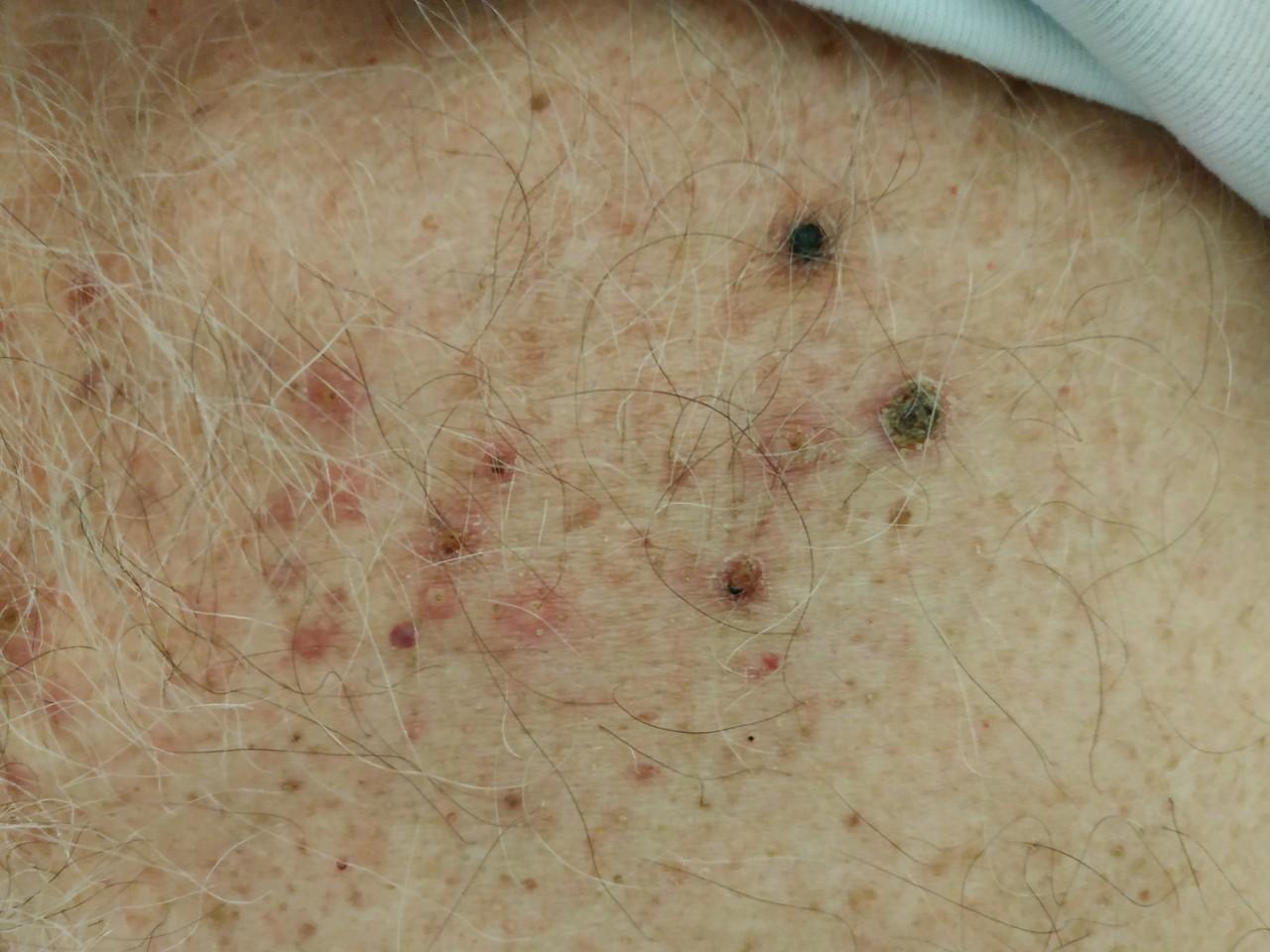
In June 2018, a 73-year-old Caucasian male was diagnosed with stage 4 colorectal adenocarcinoma with hepatic metastasis. From his past medical history, he had essential hypertension and diabetes mellitus type II. He was an ex-smoker. In July 2018, he started treatment with a combination therapy of panitumumab and folinic acid, fluorouracil and oxaliplatin (FOLFOX) every 2 weeks. After the eighth cycle of panitumumab and FOLFOX chemotherapy, the patient suddenly developed multiple, eruptive, large, sharply defined, light-brown plaques with a velvety to finely verrucous surface, mainly on the face (Fig. 1), scalp (Fig. 2) and upper trunk (Fig. 3), clinically in keeping with eruptive seborrhoeic keratosis.
Figure 1. Brownish plaques on the right preauricular and temporal area of the patient’s face
Figure 2. Several inflamed seborrhoeic keratoses on the scalp of the patient
Figure 3. Multiple eruptive seborrhoeic keratoses on erythematous bases on the upper trunk of the patient
A dermatology referral was made and the diagnosis of eruptive seborrhoeic keratosis was clinically confirmed. Upon dermatology review, the patient was advised to topically apply betamethasone 0.05% and gentamicin ointment once daily for 7 days. Panitumumab was discontinued for the ninth cycle, and 7 days later, almost all the previous new seborrhoeic keratosis resolved spontaneously. The close temporal association between panitumumab treatment and the sudden onset of multiple seborrhoeic keratoses, together with the complete recovery after 7 days of discontinuation of panitumumab treatment, pointed to a drug-related adverse event. The patient continued treatment with FOLFOX for the ninth cycle and then completed 2 more cycles of combination therapy of panitumumab and FOLFOX. The patient thereafter remained on maintenance treatment with panitumumab every 2 weeks. The patient remains to the present time under medical oncology follow-up, with no recurrence of the seborrhoeic keratosis.
DISCUSSION
Agents targeting the EGFR-mediated signalling pathway are increasingly being used for the treatment of advanced lung, pancreatic, colorectal and head and neck cancers[1]. The ability of EGFR inhibitors to block specific molecular pathways driving uncontrolled cellular division in cancer has resulted in a decreased incidence of serious systemic adverse events commonly associated with conventional cytotoxic chemotherapy[1]. However, cutaneous adverse events related to EGFR inhibitors are frequent, due to the abundant expression of EGFRs in the skin and adnexal structures[1]. Patients undergoing anti-EGFR therapy frequently present with cutaneous toxicities, which can causeserious discomfort and negatively affect compliance with anti-EGFR therapy[1,2]. Therefore, these patients may require management by dermatologists to facilitate effective oncologic management[1,2]. Cutaneous adverse reactions to EGFR inhibitors usually include acneiform (papulopustular) rash, abnormal scalp, facial hair, and/or eyelash growth, paronychia with or without pyogenic granulomas, telangiectasias, xerosis and pruritus and xerotic dermatitis[1] Interestingly, the presence and intensity of the acneiform eruptions associated with EGFR inhibitors have a positive correlation with survival[1,2]. Thus, the presence of the eruption may serve as a surrogate marker of treatment efficacy and a predictor of survival[1]. EGFR-induced multiple inflamed seborrhoeic keratoses have never been previously reported. In the epidermis, the EGFR is preferentially expressed in undifferentiated, actively proliferating basal and suprabasal keratinocytes, outer layers of the hair follicle and pilosebaceous glands[1]. Coupled to various intracellular signalling pathways, EGFRs play a critical role in maintaining epidermal homeostasis through regulation of keratinocyte proliferation, differentiation, migration and survival[2]. Exposure of epithelial cells to EGFR inhibitors induces upregulation of IL-1 and TNF-alpha and increases synthesis of other inflammatory chemokines and cytokines[3]. The theory that the inflammatory response may also be secondary to altered growth and differentiation of follicular epithelium, which results in keratotic plugging and rupture of the follicular infundibulum and influx of neutrophils, is supported by the observation that abnormal pilosebaceous units are present in both the affected and unaffected skin of patients exposed to EGFR inhibitors[3].
A wide spectrum of skin toxicities has been described in patients receiving panitumumab, an EGFR inhibitor[1], but not eruptive seborrhoeic keratosis. Although the sudden eruption of multiple seborrhoeic keratoses, called the sign of Leser-Trélat, has been reported as an ominous sign of internal malignancy[4], its validity continues to be debated. Case-control studies failed to show a significant association between eruption of numerous seborrhoeic keratoses and internal malignancies[4]. The aetiology of the Leser-Trélat sign is proposed to be the release of tumour products that act on EGFRs[5]. Only 1 case has been reported in the literature with chemotherapy-induced seborrhoeic keratosis after cytarabine treatment in a patient with acute myeloid leukaemia (AML)[5]. It is unclear whether a chemotherapy-induced eruption of seborrhoeic keratoses shares the same aetiology as the Leser-Trélat sign in patients with occult internal malignancies who have not yet been treated with chemotherapy[5].
To our knowledge, we have reported here the first case of panitumumab-induced eruptive seborrhoeic keratosis in a 73-year-old patient with metastatic colorectal cancer. While panitumumab is an emerging therapy for RAS wild-type metastatic colorectal cancer, physicians should consider panitumumab as a potential cause of eruptive seborrhoeic keratosis. The discomfort caused by such cutaneous toxicity can reduce compliance with anti-EGFR therapy. Therefore, it is important for dermatologists to be aware of and be able to effectively treat these side effects, thereby facilitating patient compliance with EGFR inhibitor therapy and playing an integral role in effective oncologic management. Although additional studies are needed in order to elucidate this further, patients may be reassured that most lesions will spontaneously resolve.