ABSTRACT
We describe the case of an 84-year-old woman who presented with right lower chest pain, anaemia and newly deranged liver function which was followed by massive upper gastrointestinal (GI) bleeding with no source of bleeding found on upper GI endoscopy. CT angiography of the GI tract confirmed rupture of a pseudoaneurysm of the right hepatic artery (RHA) that was treated successfully with trans-arterial embolization of the RHA.
LEARNING POINTS
- If upper gastrointestinal (GI) endoscopy fails to identify the source of upper GI bleeding, CT angiography is required to search for rare causes such as pseudoaneurysm of the right hepatic artery (RHA) with fistula formation with the GI and biliary tract, along with other causes such as aorto-enteric fistula.
- Pseudoaneurysm of the RHA is commonly secondary to recent surgery or trauma and spontaneous occurrence is very rare.
- Endovascular repair using transcatheter arterial embolization is the treatment of choice but if it fails, emergency laparotomy should be considered.
KEYWORDS
Pseudoaneurysm, right hepatic artery, RHA, gastrointestinal, GI, bleeding
CASE DESCRIPTION
An 84-year-old woman was brought to hospital by ambulance with acute right lower chest pain, unexplained anaemia and newly deranged liver function. The patient denied any history of cough, shortness of breath or palpitations. She had no history of venous thromboembolism (VTE) and denied risk factors for VTE. She did not have abdominal pain, vomiting, urinary symptoms, active bleeding or melena. She had normal stool habits. However, she had a history of hypertension, osteoarthritis, ischaemic heart disease with previous coronary artery bypass graft surgery many years previously, and chronic kidney disease stage 3. She was taking aspirin, and anti-hypertensive and anti-angina medication as well as a glyceryl trinitrate (GTN) spray. She had a good performance status, lived alone independently and was helped by carers only twice a week.
On examination, the patient was alert and fully conscious. Her vital signs were stable with a respiratory rate of 16 and saturating 100% on air, blood pressure of 105/59 mmHg, heart rate of 75 bpm and temperature of 36.8°C. Auscultation revealed normal heart sounds with no murmurs, and bilateral good air entry with right basal crepitations. The abdomen was soft and non-tender, bowel sounds were normal, and digital rectum examination revealed normal stool colour with no signs of fresh blood or melena. The calves were soft and non-tender and no focal neurological deficits were demonstrated.
Blood results initially showed a raised white cell count of 14×109/l (normal 4.0–10.0×109/l) and a C-reactive protein (CRP) level of 28 mg/l (0–5). Haemoglobin was 89 g/l (120–150) compared to a previous haemoglobin of 127 g/l approximately 2 months previously. Mean corpuscular volume (MCV) was 86 fl (83–101) and liver enzymes were newly deranged as follows: bilirubin 15 µmol/l (0–21), alanine aminotransferase (ALT) 304 U/l (0–33) and alkaline phosphatase (ALP) 507 U/l (30–130). We also noted urea of 11.8 mmol/l (2.5–7.8) and creatinine of 92 µmol/l (44–80). Chest x-ray showed mild haziness in the right lower zone consistent with infective changes.
Initially the patient was treated for right basal pneumonia with associated pleurisy. Because of the new anaemia, raised urea level and newly deranged liver function, she underwent urgent oesophagogastroduodenoscopy (OGD) and ultrasound of the abdomen. At this stage she was stable. OGD showed hiatus hernia and severe oesophagitis but no source of active bleeding.
Two hours later, the patient started passing a large amount of melena and her systolic blood pressure dropped to 70 mmHg despite initial intravenous fluid boluses. On arrival of the emergency team, the patient was spontaneously ventilating, on 4 l/min of oxygen saturating 98%, with good air entry bilaterally, a blood pressure of 70/48 mmHg and receiving continuous intravenous fluid resuscitation, a heart rate of 117 bpm, and a capillary refill time of 3 seconds. Blood sugars were normal and the Glasgow Coma Scale (GCS) score was 15/15. The patient was complaining of abdominal pain with a distended abdomen, generalised abdominal tenderness, involuntary guarding and sluggish bowel sounds. There were also weak peripheral pulses and no sign of VTE.
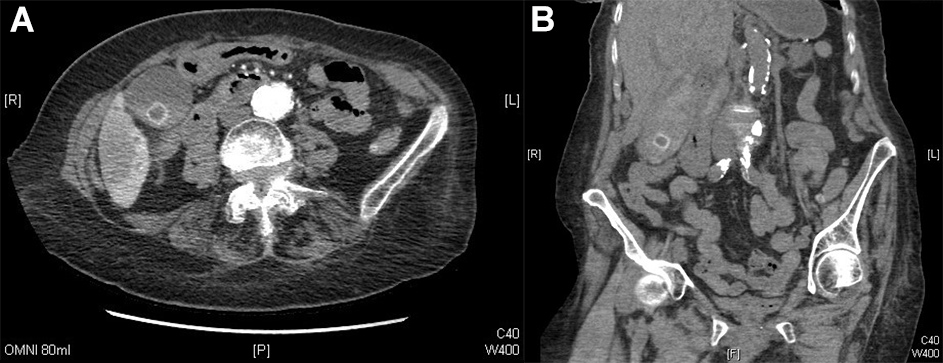
Management at this stage was for massive upper GI bleeding and included fluid resuscitation, activation of the massive haemorrhage protocol (transfusing Group O negative blood until matched bloods became available and fresh frozen plasma), proton pump inhibitor infusion, tranexamic acid 1 g and terlipressin 2 mg given on the basis of deranged liver function. A CT angiogram of the abdomen and pelvis was arranged once the patient was haemodynamically stable, and surgical review was sought for a possible perforated bowel. A CT angiogram of the abdomen (portal, venous and arterial phases) suggested a suspected pseudoaneurysm in the right hepatic artery, with surrounding fluid indicating potential active bleeding and suspected haemorrhagic content in the distended biliary tree (Fig. 1).
Figure 1. (A) A suspected pseudoaneurysm in the right hepatic artery, with surrounding fluid (likely active bleeding). (B) suspected haemorrhagic content in the distended biliary tree.
The patient was stabilised with a blood pressure of 124/78 mmHg and heart rate of 95 bpm. There was no further melena. The patient was discussed with the vascular team at the nearest tertiary centre who accepted her for transfer overnight. TAE was performed successfully and the patient was monitored for 48 hours, during which time no further signs of GI bleeding were noted. The patient improved significantly and physiologically returned to her baseline. She was discharged to the community for rehabilitation and subsequently discharged home after 2 weeks. She was followed up over the phone after 4 weeks: she was doing very well, living a normal life and reported no further chest or abdominal pains and no further GI bleeding symptoms.
DISCUSSION
Pseudoaneurysms are rare. For all RHA aneurysms (which are rare), 50% will be pseudoaneurysms[1]. Most cases are secondary to surrounding inflammation (e.g. cholecystitis and pancreatitis), recent trauma, atherosclerosis and upper GI liver or pancreatic surgery. Spontaneous rupture of a pseudoaneurysm of the RHA in the absence of any underlying cause is reported very rarely in the literature[1–3]. RHA aneurysms account for approximately 20% of all visceral aneurysms[1].
Most commonly such patients present with dull pain, a lump, jaundice and unexplained anaemia indicating sentinel bleeding (defined as a drop in haemoglobin to more than 1.5 g/dl with no signs of haemodynamic instability)[1–3]. CT angiography of the abdomen, focussing on the colic axis and superior mesenteric artery, is the gold standard diagnostic investigation. Treatment options include endovascular repair using TAE; if this fails, the next option is an emergency laparotomy[2]. The mortality rate is 50–100% in the case of rupture, making early diagnostic work-up crucial[3].